| Total CBD: | 500 – 2500 mg |
| Potency: | 16.6 - 83.3 mg/mL |
| Cost per mg CBD: | $0.12 – $0.18 |
| Extract Type: | Full-spectrum |
| THC Content: | <0.3% |
Evidence based
CBD Oil for Prion Disease (CJD & Mad Cow): What The Research Says
Prion disease is a chronic neurodegenerative disease.
There are no known cures for the condition, but recent research suggests that CBD may offer some unique benefits for treating it.
Here’s how it works.
Prion disease involves infection by a “rogue protein.”
If this protein enters the brain, it causes a chain reaction that causes the healthy protein to mutate to the same structure as the rogue version.
This is how conditions such as Creutzfeldt-Jakob Disease (CJD or “mad cow disease”) work — causing slow but widespread neurodegenerative damage over many years. The condition is eventually fatal — usually decades after the initial infection.
These infections often go unnoticed for many years. It isn’t until the condition has progressed to the point of significant damage that the condition is first diagnosed.
Does CBD work for prion disease?
The answer is inconclusive, but recent research shows CBD may be a promising treatment option in the future.

Can CBD Help With Prion Disease?
CBD has recently come to light as a potential treatment for prion disease thanks to a specific set of characteristics inherent to cannabinoids.
The fatty compounds in the cannabis plant work through a set of receptors known as the endocannabinoid receptors — tasked with regulating the balance of our hormones, neurotransmitters, and nerve function.
Cannabinoids such as CBD interact with this system to improve its ability to control processes such as dopamine production, nerve regeneration, inflammation, glutamate function, and immune cell activity in the brain — all of which are important factors involved with prion disease.
There are some aspects of CBD and other cannabinoids that suggest their use for prion disease, and preliminary research is showing promise.
However, this research is far from conclusive. We haven’t proven that it can or can’t treat prion disease yet.
As the research improves over the next couple of years, we’ll be sure to keep you updated on any breaking news.
The potential benefits of CBD for prion disease include:
- Provides widespread neuroprotective effects
- Boosts antioxidant protection
- Inhibits glutamate (NMDA) receptor activity
- Reduces brain inflammation

What is Prion Disease?
Prions are unusual disease-causing agents — they’re not bacterial, viral, or parasitic. They’re not alive — and there’s no good explanation as of yet for why they exist.
A prion is a protein. The word is derived from the proteinaceous infectious particle. Prion proteins are naturally occurring throughout the brain (PrPc), but their function isn’t well understood. Normally, these proteins are water-soluble and are broken down by enzymes known as proteases in the brain.
Diseased prions can make their way into the brain where they self-replicate by converting healthy prions into diseased ones.
These new proteins are resistant to protease, so cannot be broken down in the brain. When this happens, we start to develop “broken” proteins in the brain that we can’t get rid of. These proteins crowd areas of the brain, and since they have nothing to offer, the brain starts to decay over time.
It can take many years to begin showing symptoms.
How Does Prion Disease Work?
Initial research on prion disease discovered that the infectious agents responsible were lacking a nucleic acid genome — all previously infectious agents, including bacteria and viruses, used nucleic acids to replicate themselves.
Since prions don’t have these, it wasn’t clear how they were able to reproduce themselves in the brain.
It wasn’t until later that researchers discovered that prion diseases were caused by misshapen proteins, which would bind to other proteins and cause them to change shape as well. Prions spark a chain reaction of converting healthy proteins to unhealthy proteins that gradually spread throughout the brain — eventually leading to extensive, irreversible damage.
Where Do Prions Come From?
Before the 1950s, we had no idea what caused neurodegenerative disorders such as Creutzfeldt-Jakob Disease (CJD or “mad cow disease”).
Incidences were random, and disease progression was mysterious — leading researchers down dead ends with every attempt to understand the condition.
Sometime in the 1950s, everything changed. Researchers identified a disease among the Fore tribe in Papua New Guinea with symptoms very similar to CJD. It was identified that the disease was transmitted during a funeral ritual practiced by the tribe involving cannibalism of the brains of deceased tribe members.
Researchers noticed that some of the tribe members that were deceased had extensive brain damage (encephalopathy). Extracts made from brain samples were injected into monkeys — resulting in very similar symptoms. It was at this point that prion disease was discovered to spread through the ingestion of infected brain matter.
Other, more complex interactions may be involved as well, such as co-infection with a virus that interacts with the protein symbiotically (called the virino hypothesis), but this theory has contradictions and requires more research.
The cause and progression of prion disease are still heavily debated in the medical community, but it’s been rapidly progressing over the past five decades.
It’s now believed that prion disease starts through random mutation, which may or may not be passed on genetically. If prions from the brain get into another member of the same species, they can become infectious, propagating in the brain of those infected with the protein.

The Dangers of Prion Disease
Prion disease usually begins after eating food contaminated with the misshapen protein. In the bloodstream, the prion doesn’t pose much threat to the body, and in most cases, the body can get rid of them safely.
However, in some cases, these prions end up in places such as the spleen, peripheral neurons, or the brain, where they can cause serious long-term problems.
Ultimately, as the misshapen proteins self-replicate and spread through the brain, they kill the neurons, leaving large holes in the brain and making it look like a sponge. The scientific name for this is spongiform encephalopathy. For this reason, most prions are considered transmissible spongiform encephalopathies (TSEs).
There are a few other neurological conditions driven by misshapen proteins in the brain:
- Alzheimer’s disease
- Parkinson’s disease
- Huntington’s disease
- Spinocerebellar ataxias
- Amyotrophic lateral sclerosis (ALS)
- Diffuse Lewy body dementia
- Frontotemporal dementias
Interestingly, CBD has been shown to slow the progression of several of these neurological conditions.
Misfolded proteins in the brain are devastating in the long term, leading to gradual neurological deficits, loss of muscle control, and death.
Side Effects of Prion Disease
- Fatigue
- Difficulty speaking
- Difficulty walking and changes in gait
- Confusion
- Hallucinations
- Insomnia
- Muscle tremors
- Muscle stiffness
- Rapidly developing dementia
List of Known Human Prion Diseases
- CJD (mad cow’s disease)
- Gerstmann-Straussler-Scheinker syndrome
- Variant CJD
- Variably protease-sensitive prionopathy (VPSPr; sporadic)
- Fatal insomnia (FI)
- Kuru
Is There a Cure for Prion Disease?
Prion disease is a contractible, incurable, degenerative disease. We have drugs to manage symptoms, but currently, nothing that can cure the condition.
However, there are some anti-prion drugs in the pipeline [6]:
- Pentosan polysulfate — this is still being investigated, but there are already signs of limitation in humans.
- Anle138b — although this is still undergoing research for its effects on prion disease in humans, it is showing promising results so far.
- Astemizole — some research has shown that this compound can reduce the amount of prion protein produced by cells, thus slowing the disease progression [4].
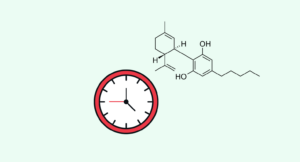
CBD can slow the progression of the disease and increase the lifespan of those affected by about 6% [3].
Suggested Reading: Is CBD Oil Safe to Use With My Medication?
There’s Limited Evidence To Support The Effects of CBD For Prion Disease
There are almost no current studies specifically investigating the effects of CBD on prion disease. So far, we’ve only found one promising study on the connection between them.
The study in question involved giving CBD to mice infected with prion disease to see how it affected their lifespan. The mice given CBD in high doses — after being infected with prion disease — had a 6% delay in death from the condition compared with the control group [3].
Researchers in this study noted that CBD prevented the accumulation of a protein known as PrPres — the hallmark sign of prion disease. They concluded that CBD could be used to increase the survival of those affected with the disorder.
However, there were some problems with this study, as pointed out by another prominent researcher.
Issues With Prion Research
1. Timing of Treatment
The mice who showed improvement with CBD use were given high doses of CBD immediately following infection. This isn’t possible when prion disease in humans isn’t diagnosed until the late stages of the condition. By the usual stage of human diagnosis, CBD application is too late.
2. Mode of Prion Infection
Another problem with this study was how the mice were infected. In the study, they were injected with the disease. This changes how the infection works in the body.
Because this disease infects the brain, a prion circulating elsewhere in the body is considered pre-infectious and will behave differently than a prion infection within the brain. Prion research reports that the body develops specific antibodies to protect itself against prions that make their way into the bloodstream [5].
3. The dose of CBD Treatment
In this study, only mice receiving high doses of CBD showed any benefit.
The effective dose used in the study was 60 mg per kg for 12 weeks. This would equate to a dose of around 4800 mg for the average male user (80 kg).
That’s 240 mL of a 600 mg CBD oil — an unreasonable amount by all standards. That’s about eight standard 30 mL bottles of CBD oil per dose.
Even if you used a CBD isolate, you’d need at least 4800 mg to match the amount used in the study — which costs around $1300 per dose. You would need this dose every day for the rest of your life since there’s no cure for prion disease currently.
Final Verdict: Using CBD for Prion Disease
Unfortunately, prion disease remains a mystery, and there’s limited research on effective treatment options for the condition. There’s only one study to date highlighting the potential role CBD may play in the progression of the disease.
However, CBD can slow the progression of other non-prion protein folding diseases such as Alzheimer’s disease [9], Parkinson’s disease [10], and Huntington’s disease [11].
The benefits of CBD come from its protective role on the neurons, lowering neuroinflammation and oxidative damage — two of the primary causes of destruction resulting from prion disease.
CBD is also useful for managing many of the side-effects of prion diseases, including:
The Future of Prion Disease: Where to Go from Here
Unfortunately, there isn’t much available research on the role of CBD or other cannabinoids for prion disease. The only study we have currently is an animal study using infected mice. The outcome of the study showed a 6% increase in lifespan among mice given high doses of CBD starting immediately after being injected with the prion [3].
The positive effects of CBD on other neurological diseases caused by misfolded proteins such Alzheimer’s disease, Huntington’s disease, and Parkinson’s disease also hint at a potential benefit from this important phytocannabinoid for prion diseases.
Recent advancements have allowed us to clone the gene responsible for manufacturing prions in mice — giving us the tools necessary to study the disease in more detail. Hopefully, there will be more research on the use of CBD for prion disease shortly.
References
- Martín-Moreno, A. M., Reigada, D., Ramírez, B. G., Mechoulam, R., Innamorato, N., Cuadrado, A., & de Ceballos, M. L. (2011). Cannabidiol and other cannabinoids reduce microglial activation in vitro and in vivo: relevance to Alzheimers′ disease. Molecular pharmacology, mol-111.
- García-Arencibia, M., González, S., de Lago, E., Ramos, J. A., Mechoulam, R., & Fernández-Ruiz, J. (2007). Evaluation of the neuroprotective effect of cannabinoids in a rat model of Parkinson’s disease: importance of antioxidant and cannabinoid receptor-independent properties. Brain research, 1134, 162-170.
- Dirikoc, S., Priola, S. A., Marella, M., Zsürger, N., & Chabry, J. (2007). Nonpsychoactive cannabidiol prevents prion accumulation and protects neurons against prion toxicity. Journal of Neuroscience, 27(36), 9537-9544.
- Karapetyan, Y. E., Sferrazza, G. F., Zhou, M., Ottenberg, G., Spicer, T., Chase, P., … & Lasmézas, C. I. (2013). Unique drug screening approach for prion diseases identifies tacrolimus and astemizole as antiprion agents. Proceedings of the National Academy of Sciences, 201303510.
- White, A. R., Enever, P., Tayebi, M., Mushens, R., Linehan, J., Brandner, S., … & Hawke, S. (2003). Monoclonal antibodies inhibit prion replication and delay the development of prion disease. Nature, 422(6927), 80.
- Trevitt, C. R., & Collinge, J. (2006). A systematic review of prion therapeutics in experimental models. Brain, 129(9), 2241-2265.
- Sailer, A., Büeler, H., Fischer, M., Aguzzi, A., & Weissmann, C. (1994). No propagation of prions in mice devoid of PrP. Cell, 77(7), 967-968.
- Langford, R. M., Mares, J., Novotna, A., Vachova, M., Novakova, I., Notcutt, W., & Ratcliffe, S. (2013). A double-blind, randomized, placebo-controlled, parallel-group study of THC/CBD oromucosal spray in combination with the existing treatment regimen, in the relief of central neuropathic pain in patients with multiple sclerosis. Journal of neurology, 260(4), 984-997.
- Esposito, G., Scuderi, C., Valenza, M., Togna, G. I., Latina, V., De Filippis, D., … & Steardo, L. (2011). Cannabidiol reduces Aβ-induced neuroinflammation and promotes hippocampal neurogenesis through PPARγ involvement. PloS one, 6(12), e28668.
- Chagas, M. H. N., Zuardi, A. W., Tumas, V., Pena-Pereira, M. A., Sobreira, E. T., Bergamaschi, M. M., … & Crippa, J. A. S. (2014). Effects of cannabidiol in the treatment of patients with Parkinson’s disease: an exploratory double-blind trial. Journal of Psychopharmacology, 28(11), 1088-1098.
- Sagredo, O., Pazos, M. R., Satta, V., Ramos, J. A., Pertwee, R. G., & Fernández‐Ruiz, J. (2011). Neuroprotective effects of phytocannabinoid‐based medicines in experimental models of Huntington’s disease. Journal of neuroscience research, 89(9), 1509-1518.
- Carter, G. T., Abood, M. E., Aggarwal, S. K., & Weiss, M. D. (2010). Cannabis and amyotrophic lateral sclerosis: hypothetical and practical applications, and a call for clinical trials. American Journal of Hospice and Palliative Medicine®, 27(5), 347-356.
More Health Benefits to Explore
-
Conditions Related to Health Benefits
- CBD For Allergies: Can This Cannabinoid Ease Symptoms?
- Top 10 CBD Oils For Back Pain
- Can CBD Help With Menstrual Cramps?
- CBD for Sciatica: How It Works, Safety, Drug Interactions, & Best Products
- Is CBD a Viable Treatment for Cerebral Palsy?
- CBD Oil For Sleep
- CBD For Psoriasis: Can CBD Help to Alleviate Symptoms?
- Traumatic Brain Injury (TBI)
- Arthritis
- Anxiety & Depression
- Weight Loss
- ADD & ADHD
- Anorexia
- Alzheimer’s Disease & Dementia
- Addiction
- ALS (Amyotrophic Lateral Sclerosis)
- Antibiotic Resistance
- Asthma
- Atherosclerosis
- Autism
- Acne
- Bipolar Disorder
- Pain
- Crohn's Disease & Ulcerative Colitis
- Diabetes
- Epilepsy
- Endocrine Disorders
- Fibromyalgia
- Fatty Liver Disease
- Glaucoma
- Hypertension
- Heart Disease
- Huntington's Disease
- Inflammation
- Irritable Bowel Syndrome (IBS)
- Kidney Disease
- Migraine Headaches
- Muscle Recovery
- Multiple Sclerosis
- Motion Sickness
- Metabolic Syndrome
- Neurodegeneration
- Cancer
- Nausea
- Neuropathic (Nerve) Pain
- Osteoporosis/Bone Health
- Obsessive-Compulsive Disorder (OCD)
- Polycystic Ovarian Syndrome (PCOS)
- PTSD
- Prion/Mad Cow Disease
- Premenstrual Syndrome (PMS)
- Parkinson’s Disease
- Schizophrenia
- Sickle Cell Anemia
- Stroke
-
Conditions Related to Products
- Ranking The Top 13 THC Gummies By Category (Δ8, Δ9, Δ10, HHC, & More)
- Top 10 CBD Oils For Back Pain
- Everything You Need to Know About CBD Sunscreen
- Top 7 CBD Gummies For Sleep & Insomnia
- Top 7 CBD Gummies To Help With Anxiety (2022)
- Best CBD Gummies For Pain (Top-Rated Pain Gummies For 2022)
- Best Hemp Cigarettes (Top 5 Nicotine-Free Smokes)
- Top 5 CBD Lip Balms For 2022
- The Top 7 CBD Face Masks for 2022
- The Best CBD Inhalers For 2022 (& How to Use Them)
- Best Full-Spectrum CBD Vape Juice: What to Look For & How to Use It
- CBD Eye Drops: New Option For Glaucoma?
- CBD Oil For Dogs With Arthritis
- Best CBD Massage Oils In 2022
- Buyer's Guide To The Best CBD Vape Kits In 2022
- CBD Chocolate: Yes, It Exists & It's Just as Divine as it Sounds
- CBD Pre-Rolls & Cigarettes
- Terpene Concentrates
- Best CBD Soaps
- Best CBD Shampoo & Conditioner
- Best CBD Juul Pods
- CBD Isolate Oils
- Full-Spectrum CBD Oils
- Best CBD Lube
- CBD Honey
- CBD Transdermal Patches
- Best Dry Herb Vaporizers
- CBD Oil For Dogs With Epilepsy
- CBD Oil For Dogs With Anxiety
- CBD Oil For Dogs With Cancer
- CBD For Horses
- CBD Chewing Gum
- CBD Pain Cream
- CBD Oil For Cats
- CBD Oil For Dogs
- CBD Hemp Flower
- CBD Suppositories
- Best CBD Gummies for Pain, Sleep & Anxiety Reviewed (2022)
- CBD Teas
- CBD Vape Pens
- CBD Vape Oils
- CBD Coffee
- CBD Drinks & Shots
- CBD Crystals
- CBD Skincare
- Best CBD Oil & Gummies For Kids: Is CBD Safe for Children with Anxiety & ADHD?
- CBD Concentrates
- CBD Bath Bombs
- CBD Capsules
- CBD Sprays
- CBD Dog Treats
-
Conditions Related to Topicals
-
Conditions Related to Oils & Tinctures
-
Conditions Related to Edibles
- Top 7 CBD Gummies To Help With Anxiety (2022)
- Best CBD Gummies For Pain (Top-Rated Pain Gummies For 2022)
- CBD Chocolate: Yes, It Exists & It's Just as Divine as it Sounds
- CBD Honey
- CBD Chewing Gum
- Best CBD Gummies for Pain, Sleep & Anxiety Reviewed (2022)
- CBD Teas
- CBD Coffee
- CBD Drinks & Shots
- CBD Capsules
-
Conditions Related to Gummies
- Ranking The Top 13 THC Gummies By Category (Δ8, Δ9, Δ10, HHC, & More)
- Top 7 CBD Gummies For Sleep & Insomnia
- Top 7 CBD Gummies To Help With Anxiety (2022)
- Best CBD Gummies For Pain (Top-Rated Pain Gummies For 2022)
- Best CBD Gummies for Pain, Sleep & Anxiety Reviewed (2022)
- Best CBD Oil & Gummies For Kids: Is CBD Safe for Children with Anxiety & ADHD?
-
Conditions Related to Hemp Flower
-
-
Conditions Related to Terpenes
-
-
Conditions Related to Cultivation
-
Conditions Related to Concentrates
-
Conditions Related to Delta 8 THC
-
Conditions Related to Delta 9 THC
-
-
-
-
Conditions Related to CBD
- Everything You Need to Know About CBD Sunscreen
- Top 7 CBD Gummies For Sleep & Insomnia
- Top 7 CBD Gummies To Help With Anxiety (2022)
- Best CBD Gummies For Pain (Top-Rated Pain Gummies For 2022)
- Best Hemp Cigarettes (Top 5 Nicotine-Free Smokes)
- Top 5 CBD Lip Balms For 2022
- The Top 7 CBD Face Masks for 2022
- The Best CBD Inhalers For 2022 (& How to Use Them)
- Best Full-Spectrum CBD Vape Juice: What to Look For & How to Use It
- CBD Eye Drops: New Option For Glaucoma?
- CBD Oil For Dogs With Arthritis
- Best CBD Massage Oils In 2022
- Buyer's Guide To The Best CBD Vape Kits In 2022
- CBD Chocolate: Yes, It Exists & It's Just as Divine as it Sounds
- CBD Pre-Rolls & Cigarettes
- Best CBD Soaps
- Best CBD Shampoo & Conditioner
- Best CBD Juul Pods
- CBD Isolate Oils
- Full-Spectrum CBD Oils
- Best CBD Lube
- CBD Honey
- CBD Transdermal Patches
- CBD Oil For Dogs With Epilepsy
- CBD Oil For Dogs With Anxiety
- CBD Oil For Dogs With Cancer
- CBD For Horses
- CBD Chewing Gum
- CBD Pain Cream
- CBD Oil For Cats
- CBD Oil For Dogs
- CBD Hemp Flower
- CBD Suppositories
- Best CBD Gummies for Pain, Sleep & Anxiety Reviewed (2022)
- CBD Teas
- CBD Vape Pens
- CBD Vape Oils
- CBD Coffee
- CBD Drinks & Shots
- CBD Crystals
- CBD Skincare
- Best CBD Oil & Gummies For Kids: Is CBD Safe for Children with Anxiety & ADHD?
- CBD Concentrates
- CBD Bath Bombs
- CBD Capsules
- CBD Sprays
- CBD Dog Treats
-
-
Conditions Related to THC-O
-
-
Conditions Related to Joint Health
-
Conditions Related to Pain Disorders
- Top 10 CBD Oils For Back Pain
- Can CBD Help With Menstrual Cramps?
- CBD for Sciatica: How It Works, Safety, Drug Interactions, & Best Products
- Traumatic Brain Injury (TBI)
- Arthritis
- Pain
- Fibromyalgia
- Glaucoma
- Inflammation
- Kidney Disease
- Migraine Headaches
- Multiple Sclerosis
- Neuropathic (Nerve) Pain
- Premenstrual Syndrome (PMS)
- Sickle Cell Anemia
-
Conditions Related to Autoimmune Disease
-
Conditions Related to Cognitive Health
-
Conditions Related to Metabolic Disorders
-
Conditions Related to Psychological Disorders
-
Conditions Related to Muscles & Bones
-
Conditions Related to Nervous System
- CBD for Sciatica: How It Works, Safety, Drug Interactions, & Best Products
- Is CBD a Viable Treatment for Cerebral Palsy?
- CBD Oil For Sleep
- Traumatic Brain Injury (TBI)
- Anxiety & Depression
- ADD & ADHD
- Anorexia
- Alzheimer’s Disease & Dementia
- Addiction
- ALS (Amyotrophic Lateral Sclerosis)
- Autism
- Bipolar Disorder
- Epilepsy
- Huntington's Disease
- Inflammation
- Migraine Headaches
- Multiple Sclerosis
- Motion Sickness
- Neurodegeneration
- Neuropathic (Nerve) Pain
- Obsessive-Compulsive Disorder (OCD)
- PTSD
- Prion/Mad Cow Disease
- Parkinson’s Disease
- Schizophrenia
-
-
Conditions Related to Reproductive Health
-
Conditions Related to Hormones & Endocrine
-
Conditions Related to Skin Health
-
Conditions Related to Cardiovascular System
-
Conditions Related to Digestive System
-
Conditions Related to Genetic Disorders
-
Conditions Related to For Children