Evidence based
How Are Cannabinoids Metabolized? (CBD, THC, CBG, THC-O, & More)
What happens to cannabinoids after they enter your body? Where do they go?
Cannabis contains over 421 individual ingredients spread across 18 different classes of chemicals.
The vast majority of these compounds require some form of metabolism for them to be eliminated from the body. Scientists are still trying to understand exactly how this process works, but have uncovered some interesting findings in the process.
Here, we’ll explain how cannabinoids like CBD and THC are metabolized and how this may affect the way these compounds interact with your other medications and supplements.
Drug Metabolism 101
Almost all drugs we consume need to be metabolized before they can be eliminated from the body.
There are two main phases of drug metabolism that are relevant when it comes to cannabinoids.
A) Phase-I Metabolism (CYP450)
Phase I metabolism (oxidation, reduction, hydrolysis, cyclization) is the first step in metabolizing drugs (including cannabinoids). These reactions rely on a series of specialized enzymes known collectively as the cytochrome P450 monooxidase system (CYP450 for short).
There are more than 300,000 distinct CYP proteins throughout all kingdoms of life. Plants, animals, bacteria, fungi, and even a few viruses use CYP enzymes to metabolize compounds from their environment.
In humans, the CYP enzymes are responsible for around 75% of total metabolism in the human body [15]. They’re also responsible for both synthesizing and metabolizing hormones, such as glucocorticoids, estrogens, testosterone, and more.
In some cases, phase I metabolism deactivates drugs; in other cases, it makes them more active (as is the case with prodrugs like THC-O, codeine, or 1P-LSD).
For cannabinoids, phase I metabolism produces both active and inactive metabolites. For example, 11-OH-THC is an active metabolite of THC, while THC-COOH is an inactive metabolite. Both are formed via phase I metabolism.
B) Phase II Metabolism (Conjugation)
Phase II metabolism (conjugation) involves the addition of another functional group to the metabolites formed during phase I. Common additions include glutathione, sulfate, glycine, or glucuronic acid.
In the metabolism of cannabinoids, glucuronic acid is the primary conjugation species used. The final byproducts of THC and CBD are both glucuronic acid conjugates (THC-COOH-Glucuronic Acid and CBD-COOH-Glucuronic acid, respectively).
The purpose of phase II metabolism is to deactivate compounds and increase both water solubility and renal clearance. This helps the body eliminate the drug from the body through either the kidneys, the feces, or both.
How is CBD Metabolized?
There are more than 30 identified metabolites of CBD (cannabidiol) [11]. However, the primary metabolite is 7-hydroxy-CBD (7-OH-CBD) and 7-COOH-CBD — both of which are considered just as potent as CBD itself [16].
Phase I metabolism of CBD primarily involves the CYP3A4 [13], CYP2C19, and CYP2D6 enzymes [14].
Other metabolites include 7-COOH-CBD, 6α-OH-CBD, 6β-OH-CBD, 2-OH-CBD, 4-OH-CBD, 5-OH-CBD, 3-OH-CBD, and 1-OH-CBD [17].
Next, 7-COOH-CBD is converted into various glucuronide conjugates via phase II liver metabolism before being excreted via the urine and feces.
Most of the phase II metabolism of CBD is carried out by the UDP-glucuronosyltransferases UGT1A9 and UGT2B7 [14].
What’s The Half-Life of CBD?
The half-life of CBD is around 1–2 days for infrequent users [18] and up to 5 days for heavy users (classified as taking more than 10 mg per day for at least 2-weeks straight) [19].
This means one dose of CBD can remain detectable in the body for up to 2 weeks.
CBD remains longer in the bodies of those who take CBD on a daily basis compared to those who use CBD every once in a while.
How is THC Metabolized?
THC metabolism begins immediately, with some metabolism even occurring directly in the lungs within a few seconds of smoking or vaping marijuana (side-chain hydroxylation).
Phase I metabolism of THC involves hydroxylation and oxidation via the CYP2C9, CYP2C19, and CYP3A4 isoenzymes in the liver [4].
There are more than 100 metabolites of THC [3], but the primary metabolites are 11-hydroxytetrahydrocannabinol (11-OH-THC) and 11-nor-9-carboxy-tetrahydrocannabinol (THC-COOH) [1,2].
11-OH-THC is an active metabolite (provides psychoactive effects). Concentrations of this compound peak roughly 13 min after smoking [5].
The second primary metabolite, THC-COOH, is inactive, which means it carries no psychoactive effects. This compound forms as a result of further oxidation of 11-OH-THC. Peak levels of this compound appear around 81 minutes after smoking marijuana [6].
Next, both metabolites are converted to 8α-OH-THC, THC-COOH-glucuronide, and other glucuronide conjugates through phase II liver metabolism. During this stage, glucuronic acid is added to THC-COOH, which improves water solubility and facilitates elimination via the kidneys.
How is Delta 8 THC Metabolized?
Delta 8 THC follows the same pathways of elimination as delta 9 THC. It’s also converted to 7-OH-THC and THC-COOH before being conjugated and eliminated via the bile and urine.
Other isomers of THC likely follow the same metabolic pathways — including delta 10 THC, HHC, and THCP — however, there’s yet to be any significant studies examining the metabolites of these relatively “new” cannabinoids.
What is the Half-Life of THC?
The half-life of THC is about 1.3 days for infrequent users and around 5–13 days for frequent users [8].
One of the primary metabolites of THC, THC-COOH, remained detectable in the blood for up to 74.3 hours [9].
How is THC Excreted?
More than 65% of cannabis is excreted in the feces, and approximately 20% is excreted in urine [11]. Some THC is eliminated in its initial form, but most of it is converted to 7-OH-THC, THC-COOH, and various THC glucuronide conjugates first.
How is THCV Metabolized?
Unfortunately, there aren’t any solid studies examining the full metabolic profile of THCV. However, as a homolog of THC, THCV likely follows the same metabolic pathways as THC.
Some research has suggested THCV-COOH and its conjugates as the primary metabolites of THCV in urine [20]. These metabolites were reportedly detected for up to 2-weeks after smoking just one marijuana cigarette.
How is CBG Metabolized?
CBG (cannabigerol) metabolism still isn’t very well understood. The best studies we have exploring this cannabinoid come from the early 90s.
From the research currently available, it appears CBG follows the same metabolic pathways as THC and CBD — first undergoing phase I oxidation reactions, followed by the formation of glucuronide conjugates before being excreted by both the kidneys and bile [21,22].
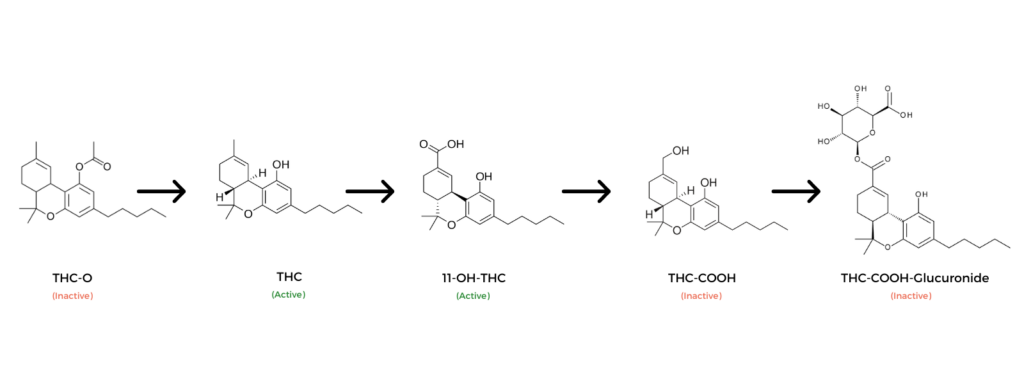
How Is THC-O Metabolized?
THC-O (aka THCO) is a prodrug for THC. This means the body converts THC-O into THC through the same metabolic pathways used to break down other cannabinoids. This is a prime example of a case where metabolism actually makes a drug stronger rather than neutralizing it.
THC-O is made by esterifying an acetate group to THC or THCA. This can be done to all forms of THC, including delta 8 THC, delta 9 THC, and delta 10 THC.
The addition of an acetate group significantly enhances the absorption of the cannabinoid. Once in the body, it’s converted to either delta 8, delta 9, or delta 10 THC.
From here, the THC is metabolized to 7-OH-THC, THC-COOH, and end conjugate products.
Because of the extra step of activating THC-O, this compound takes longer to kick in (roughly 1.5 hours) and remains active for up to 25% longer than conventional THC.
There are also other esterified versions of THC, such as THC-O-phosphate, THC hemisuccinate, and THC morpholinylbutyrate — however, none of these compounds have received the same attention or popularity as THC-O.
What Factors Affect Cannabinoid Metabolism?
Drug metabolism is complicated. There are dozens of factors that can affect how fast or efficiently a given cannabinoid (or drug) will be metabolized by an individual.
The main factors that can affect the metabolism of cannabinoids include:
- Age — metabolism tends to slow down as we age.
- Other medications — if other medications or substances compete for the same pathways, it can slow down our ability to metabolize cannabinoids.
- Genetic differences — individual genetics can significantly affect the way we produce CYP450 enzymes and alter our ability to metabolize different kinds of drugs.
- Underlying illness — liver disease can slow the body’s ability to metabolize drugs. Kidney disease can interfere with the ability to eliminate these metabolites from the bloodstream.
Do Cannabinoids Interact With Other Medications?
Yes, cannabinoids such as CBD can interact with other medications by competing for individual metabolic pathways. When two or more compounds require the same enzymes for metabolism, they’re forced to wait for an available slot in order to be processed. This causes metabolism to slow down.
Cannabinoids can interact in other ways, too — such as increasing the risk of side effects (such as hypotensive medications) or negating their effectiveness (such as immunosuppressants or stimulant medications).
How Metabolic Interaction Works
Let’s use the drive-through as a metaphor for this.
Say you’re going to grab some food in your car. Tonight, you’re craving some McDonalds, so you head over to the drive-through. You’re the only one there, so you rush through with your food and return home within 15 minutes.
On another night, you head out to the same McDonalds, but it’s a Friday, and nobody else feels like cooking either. When you arrive, there are already about a dozen other cars in line ahead of you. You’re going to have to wait for the restaurant to process each order one at a time, so by the time you manage to place your order, pay, pick up your meal, and head home, it’s been a full hour.
Drug metabolism works in a similar way. If there’s a lot of demand for one particular CYP450 enzyme group, it’s going to take longer to process each compound individually.

How Serious Are Metabolic Interactions With Cannabinoids & Prescription Medications?
The risk of negative interactions when using non-psychoactive cannabinoids, such as CBD, CBG, or CBC, are relatively low.
Metabolic competition with cannabinoids can become an issue with drugs that require careful titration or rely heavily on CYP450 metabolism.
For example, warfarin, a common blood thinner, requires very careful titration to be safe and effective. This means the amount of the drug consumed needs to be carefully balanced with the rate of metabolism. If the metabolic rate is slowed down for any reason, warfarin can begin to accumulate in the blood. Taking cannabinoids (or other medications or supplements) alongside warfarin can (theoretically) slow metabolism and cause it to build up to unsafe levels in the body.
Which Drugs Aren’t Safe to Take Alongside Cannabinoids?
High-Risk Metabolic Interactions With Cannabinoids:
This group carries the highest risk of interaction, as well as the most severe side effects. These medications are commonly used on a regular basis and have a narrow therapeutic window.
Taking these medications alongside CBD, THC, or other cannabinoids on a recurring basis can cause serum levels to rise, eventually leading to side effects of toxicity.
| Drug Name | Trade Names | Enzymes | Drug Class |
| Alprazolam | Xanax, Xanor, Niravam | CYP3A4 | Benzodiazepine |
| Clonazepam | Klonopin | CYP3A4 | Benzodiazepine |
| Cyclophosphamide | Cytoxan, Neosar | CYP2B6, CYP3A4, CYP2C9 | Immunosuppressant |
| Cyclosporine | Gengraf,Sandimmune,Neoral | CYP3A4 | Immunosuppressant |
| Diazepam | Valium | CYP3A4, CYP2C19, 21-Hydroxylase | Benzodiazepine |
| Estazolam | Prosom | CYP3A4 | Benzodiazepine |
| Ketoconazole | Nizoral | CYP2A6, CYP2C19, CYP3A4 | Imidazoles |
| Methotrexate | Rheumatrex, Trexall | CYP3A4, CYP3A5 | Immunosuppressant |
| Mycophenolate mofetil | Cellcept | CYP3A4 | Immunosuppressant |
| Warfarin | Coumadin,Coupadin,Jantoven | CYP3A4, CYP2C9 | Blood Thinner |
Moderate Risk Interactions With Cannabinoids:
These drugs rely heavily on CYP3A4 and CYP2D6 for metabolism and are used on a daily basis.
Compared to the high-risk group, these drugs are less likely to interact with cannabinoids, even with daily use. However, caution is advised when taking any prescription medication alongside cannabinoids. There are other types of interactions that can occur apart from metabolic competition.
| Name | Trade Names | CYP450 Targets | Drug Class |
| Amitriptyline | Elavil, Endep, Vanatrip | CYP2C19, CYP2D6 | Tricyclic Antidepressant |
| Amlodipine | Norvasc | CYP2A6, CYP3A4 | Calcium Channel Blockers |
| Amlodipine besylate | Lotrel, Norvasc | CYP3A4 | Calcium Channel Blockers |
| Amphetamine | Adzenys ER, Adderall, Mydayis | CYP2D6 | Stimulants |
| Aripiprazole | Abilify | CYP2D6, CYP3A4 | SDA Medications |
| Atorvastatin | Lipitor | CYP3A4 | Statin |
| Caffeine | Lucidex, Vivarin, NoDoz, Revive | CYP1A2, CYP2E1, CYP3A4, CYP2C8, CYP2C9 | Stimulants |
| Captopril | Capoten | CYP2D6 | Ace Inhibitors |
| Carvedilol | Coreg | CYP2D6, CYP1A2, CYP3A4 | Alpha-Blocker |
| Chloramphenicol | Chloromyectin | CYP2C9, CYP2C19, CYP3A4 | Antibiotic |
| Citalopram | Celexa, Cipramil | CYP2C19, CYP3A4, CYP2D6, CYP1A1 | SSRI |
| Cocaine | Goprelto, Numbrino, Neurocaine | CYP2D6 | Stimulants |
| Dexamethasone | DexPak, Decadron | CYP2D6 | Anti-Emetic |
| Diltiazem | Cardizem, Dilacor XR, Cartia XT | CYP3A4, CYP2D6 | Calcium Channel Blockers |
| Diphenhydramine | Benadryl, Unisom, Nytol | CYP2D6, CYP1A2, CYP2C9, CYP2C19 | Antihistamine |
| Doxepin | Aponal, Quitaxon, Sinequan | CYP2C19, CYP2D6, CYP1A2 | Tricyclic Antidepressant |
| Duloxetine | Cymbalta, Ariclaim, Yentreve | CYP2D6, CYP1A2 | SNRI |
| Escitalopram | Cipralex, Lexapro | CYP2C19, CYP2D6, CYP3A4 | SSRI |
| Fluoxetine | Prozac, Rapiflux, Sarafem, Selfemra, Oxactin | CYP2C19, CYP2D6 | SSRI |
| Fluvoxamine | Faverin | CYP2D6, CYP1A2, CYP1A1, CYP2C9, CYP2C19, CYP3A4 | SSRI |
| Hydroxyzine | Atarax, Vistaril | CYP2D6 | Antihistamine |
| Metoclopramide | Reglan, Metozolv ODT | CYP2D6, CYP1A2 | Anti-Emetic |
| Modafinil | Provigil, Nuvigil | CYP2A6, CYP2C9, CYP2C19, CYP1A1, CYP3A4 | Stimulants |
| Nifedipine | Procardia XL, Adalat CC | CYP3A4 | Calcium Channel Blockers |
| Nortriptyline | Pamelor, Aventyl HCl | CYP2D6, CYP1A2, CYP2C19, CYP3A4 | Tricyclic Antidepressant |
| Olanzapine | Zyprexa | CYP1A2, CYP2D6 | SDA Medications |
| Ondansetron | Zofran, Zuplenz, Ondisolv | CYP3A4, CYP2D6, CYP1A2 | Anti-Emetic |
| Paroxetine | Seroxat, Paxil, Paxil CR, Pexeva | CYP2C9, CYP2D6 | SSRI |
| Perindopril | Aceon | CYP3A4 | Ace Inhibitors |
| Prochlorperazine | Compazine | CYP2D6 | Anti-Emetic |
| Promethazine | Phenergan | CYP2C9, CYP2D6 | Antihistamine |
| Quetiapine | Seroquel, Temprolide | CYP3A4 | SDA Medications |
| Ramipril | Altace | CYP3A4 | Ace Inhibitors |
| Sertraline | Zoloft, Lustral | CYP2B6, CYP2C19, CYP2C9, CYP3A4, CYP2D6 | SSRI |
| Simvastatin | Zocor | CYP3A4 | Statin |
| Verapamil | Calan SR, Verelan, Isoptin SR, Covera-HS | CYP1A1, CYP3A4 | Calcium Channel Blockers |
Summary: How Are Cannabinoids Metabolized?
Most cannabinoids follow the same pathways of metabolization in the human body.
Metabolism begins immediately — starting in the lungs for smoked cannabis and in the small intestines for CBD consumed orally,.
However, the bulk of metabolism happens later; once the cannabinoids reach the liver. Here, a group of enzymes collectively referred to as the CYP450 complex breaks each cannabinoid down further. THC is converted to 7-OH-THC, which remains psychoactive, before breaking down into THC-COOH and smaller conjugated metabolites (which are inactive).
CBD follows a similar path — first, it’s converted to 7-OH-CBD, then 7-COOH-CBD, and finally smaller conjugates which are able to be filtered and eliminated by the kidneys and bile.
Understanding the way cannabinoids are metabolized is essential for determining the most effective doses and for spotting negative interactions with other drugs before they cause any physical harm.
References
- Huestis, M. A. (2007). Human cannabinoid pharmacokinetics. Chemistry & biodiversity, 4(8), 1770.
- Claussen, U., & Korte, F. (1968). Concerning the behavior of hemp and of delta-9-6a, 10a-trans-tetrahydrocannabinol in smoking. Justus Liebigs Annalen der Chemie, 713, 162-165.
- Grotenhermen, F. (2003). Pharmacokinetics and pharmacodynamics of cannabinoids. Clinical pharmacokinetics, 42(4), 327-360.
- Matsunaga, T., Iwawaki, Y., Watanabe, K., Yamamoto, I., Kageyama, T., & Yoshimura, H. (1995). Metabolism of Δ9-tetrahydrocannabinol by cytochrome P450 isozymes purified from hepatic microsomes of monkeys. Life sciences, 56(23-24), 2089-2095.
- Huestis, M. A., Henningfield, J. E., & Cone, E. J. (1992). Blood cannabinoids. I. Absorption of THC and formation of 11-OH-THC and THCCOOH during and after smoking marijuana. Journal of analytical Toxicology, 16(5), 276-282.
- Huestis, M. A. (2007). Human cannabinoid pharmacokinetics. Chemistry & biodiversity, 4(8), 1770.
- Krishna, D. R., & Klotz, U. (1994). Extrahepatic metabolism of drugs in humans. Clinical pharmacokinetics, 26(2), 144-160.
- Smith-Kielland, A., Skuterud, B., & Mørland, J. (1999). Urinary excretion of 11-nor-9-carboxy-Δ9-tetrahydrocannabinol and cannabinoids in frequent and infrequent drug users. Journal of Analytical Toxicology, 23(5), 323-332.
- Reiter, A., Hake, J., Meissner, C., Rohwer, J., Friedrich, H. J., & Oehmichen, M. (2001). Time of drug elimination in chronic drug abusers: Case study of 52 patients in a “low-step” detoxification ward. Forensic Science International, 119(2), 248-253.
- Karschner, E. L., Schwilke, E. W., Lowe, R. H., Darwin, W. D., Herning, R. I., Cadet, J. L., & Huestis, M. A. (2009). Implications of plasma Δ9-tetrahydrocannabinol, 11-hydroxy-THC, and 11-nor-9-carboxy-THC concentrations in chronic cannabis smokers. Journal of analytical toxicology, 33(8), 469-477.
- Lemberger, L., Axelrod, J., & Kopin, I. J. (1971). Metabolism and disposition of Δ9-tetrahydrocannabinol in man. Pharmacological Reviews, 23(4), 371-380.
- Millar, S. A., Stone, N. L., Yates, A. S., & O’sullivan, S. E. (2018). A systematic review on the pharmacokinetics of cannabidiol in humans. Frontiers in pharmacology, 9, 1365.
- Watanabe, K., Usami, N., Osada, S., Narimatsu, S., Yamamoto, I., & Yoshimura, H. (2019). Cannabidiol metabolism revisited: tentative identification of novel decarbonylated metabolites of cannabidiol formed by human liver microsomes and recombinant cytochrome P450 3A4. Forensic Toxicology, 37(2), 449-455.
- Al Saabi, A., Allorge, D., Sauvage, F. L., Tournel, G., Gaulier, J. M., Marquet, P., & Picard, N. (2013). Involvement of UDP-glucuronosyltransferases UGT1A9 and UGT2B7 in ethanol glucuronidation and interactions with common drugs of abuse. Drug Metabolism and Disposition, 41(3), 568-574.
- Guengerich, F. P. (2008). Cytochrome p450 and chemical toxicology. Chemical research in toxicology, 21(1), 70-83.
- Beers, J. L., Fu, D., & Jackson, K. D. (2021). Cytochrome P450–Catalyzed Metabolism of Cannabidiol to the Active Metabolite 7-Hydroxy-Cannabidiol. Drug Metabolism and Disposition, 49(10), 882-891.
- Vlad, R. A., Hancu, G., Ciurba, A., Antonoaea, P., Rédai, E. M., Todoran, N., … & Muntean, D. L. (2020). Cannabidiol-therapeutic and legal aspects. Die Pharmazie-An International Journal of Pharmaceutical Sciences, 75(10), 463-469.
- Welty, T. E., Luebke, A., & Gidal, B. E. (2014). Cannabidiol: Promise and Pitfalls: Cannabidiol: Promise and Pitfalls. Epilepsy currents, 14(5), 250-252.
- Consroe, P., Kennedy, K., & Schram, K. (1991). Assay of plasma cannabidiol by capillary gas chromatography/ion trap mass spectroscopy following high-dose repeated daily oral administration in humans. Pharmacology Biochemistry and Behavior, 40(3), 517-522.
- Levin, F. R., Mariani, J. J., Brooks, D. J., Xie, S., & Murray, K. A. (2010). Δ9-Tetrahydrocannabivarin testing may not have the sensitivity to detect marijuana use among individuals ingesting dronabinol. Drug and alcohol dependence, 106(1), 65-68.
- Harvey, D. J., & Brown, N. K. (1990). In vitro metabolism of cannabigerol in several mammalian species. Biomedical & environmental mass spectrometry, 19(9), 545-553.
- Hidvegi, E., & Somogyi, G. P. (2010). Detection of cannabigerol and its presumptive metabolite in human urine after Cannabis consumption. Die Pharmazie-An International Journal of Pharmaceutical Sciences, 65(6), 408-411.