What Are Cannabinoids?
Cannabinoids like CBD contribute most of the therapeutic effects of the cannabis plant.
Learn how they work & why they’re important.
CBD | CBG | CBN | THC | & More
There are hundreds of compounds produced in the cannabis plant.
However, most of the therapeutic action comes down to just two groups of compounds — the cannabinoids and the terpenes.
In this article, we’ll cover everything you need to know about cannabinoids.
We’ll cover phytocannabinoids from hemp (such as CBD, CBG, CBC, THCV), endocannabinoids produced in the body (anandamide & 2-AG), and synthetic or pharmaceutical versions being used as illicit drugs and medicine.

What Are Cannabinoids?
A cannabinoid is any compound, natural or synthetic, that interacts with the endocannabinoid system (ECS).
The ECS is a system found in all mammals, reptiles, and fish.
This system is very complex, and scientists are still seeking to understand how it works. So far, the ECS has been found to play a role in regulating hormones, mood, appetite, sleep, memory, reproductive function, and much more.
There are three main types of cannabinoids:
- Phytocannabinoids — cannabinoids made by plants
- Endocannabinoids — cannabinoids naturally made by the body
- Synthetic Cannabinoids — man-made cannabinoids
When talking about cannabinoids, most people refer specifically to the compounds produced in cannabis. There are a total of 144 individual cannabinoids discovered in the cannabis plant so far, with new ones being discovered every couple of years [1].
The most predominant cannabinoid in marijuana is delta 9 THC, which is the active ingredient that makes users high.
In the hemp plant, CBD is much more predominant and is considered the primary active ingredient in terms of the plant’s health benefits. This cannabinoid is completely non-psychoactive (won’t make you high) and offers an impressive range of health benefits.
CBD doesn’t work alone. There are dozens of other cannabinoids in the plant that offer their own set of medicinal benefits as well.
Cannabinoid Benefits Chart
| Health Benefit | CBD | THC | CBN | CBG | CBC | THCV |
| Alleviates Pain | ✓ | ✓ | ✓ | ✓ | ||
| Reduces Inflammation | ✓ | ✓ | ✓ | ✓ | ||
| Prevents Acne | ✓ | ✓ | ✓ | |||
| Protects the Brain | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Regulates Mood | ✓ | ✓ | ✓ | |||
| Alleviates Epilepsy | ✓ | |||||
| Relieves Anxiety | ✓ | ✓ | ||||
| Stimulates Appetite | ✓ | ✓ | ||||
| Inhibits Appetite | ✓ | ✓ | ✓ | |||
| Fights Cancer | ✓ | ✓ | ✓ | ✓ | ||
| Alleviates Glaucoma Symptoms | ✓ | ✓ | ✓ | |||
| Supports Sleep | ✓ | ✓ | ✓ | ✓ |
I. Phytocannabinoids
A phytocannabinoid is a compound that mimics the effects or structure of cannabinoids naturally produced by the body (endocannabinoids). They activate or block the receptors within the endocannabinoid system.
The cannabis plant is the largest natural source of phytocannabinoids. It contains around 144 individual compounds, each one with a unique impact on the body.
Some phytocannabinoids stimulate ECS receptors, others inhibit.
CBD is unique because it works as a regulator — acting to both increase and decrease ECS activity to achieve balance in the system overall.
This section will break down the most common phytocannabinoids produced in the cannabis plant, including how they work and what benefits they provide.
1. CBD (Cannabidiol)
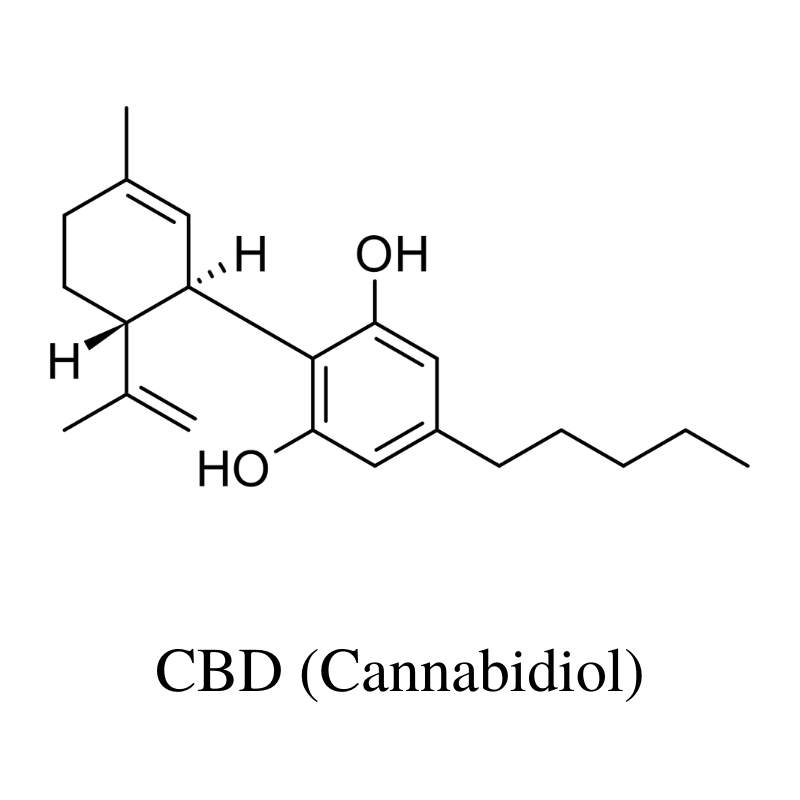
CBD (cannabidiol) is the primary cannabinoid in the hemp plant. It’s completely non-psychoactive (doesn’t make you high) and provides a long list of health benefits through its ability to regulate the ECS.
This is considered the active ingredient in CBD oils and is the most abundant by far.
There is a ton of research available exploring the effects of CBD and there are even several pharmaceutical products using CBD as the active ingredient for treating a variety of different health conditions. While CBD has many benefits, the most predominant is its ability to block pain, inhibit inflammation, and promote a relaxed mental state.
The benefits of CBD include:
- Alleviate pain & inflammation
- Support mental health & wellbeing
- Support healthy skin
- Protect the brain
- Support digestive function
- May protect the cardiovascular system
- Support reproductive health
- Regulate hormones & metabolism
How CBD Works:
CBD works by regulating the endocannabinoid system. The interaction is very complex — researchers are still trying to understand exactly how this unique cannabinoid actually works.
From a biochemical perspective, CBD binds to the ECS receptors (both CB1 and CB2), inhibits GPR55 receptors, and stimulates serotonin 5-HT1A receptors [2].
CBD has also been shown to interact with a variety of other receptors — including adenosine (reuptake inhibitor), PPARγ nuclear receptor (stimulates), TRPV1 receptors (stimulates), Mg2+‐ATPase (inhibits), 15‐lipoxygenase (inhibits), and much more.
CBD vs. CBDa
The natural form of CBD is CBDa — cannabidiolic-acid. The acid group breaks off when it’s exposed to heat or oxygen. Raw CBD products often contain high concentrations of CBDa, while processed CBD products are higher in CBD.
Both versions have very similar effects on the body. There’s some evidence to suggest that CBDa has a stronger effect on the body, but the research is limited. The vast majority of CBD research uses the decarboxylated from — CBD.
2. CBDV (Cannabidivarin)
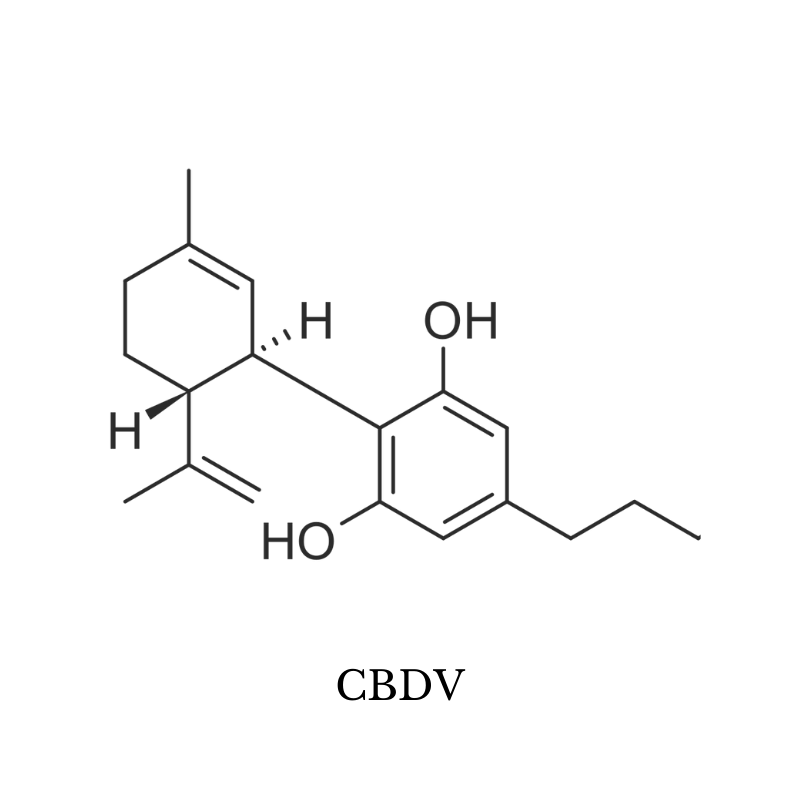
CBDV (cannabidivarin) is structurally very similar to CBD and shares many of the same benefits.
The concentration of CBDV in cannabis plants is very low — categorizing it as a minor cannabinoid.
There isn’t much research on CBDV yet, but things are about to change. GW Pharmaceuticals, the company that makes an FDA-approved CBD drug called Epidiolex, has been developing a new drug based on the CBDV molecule called GPW42006. The company has been conducting several large-scale clinical trials on the effects of CBDV for treating epilepsy.
Animal testing has shown CBDV may inhibit acne formation by controlling the amount of sebum produced by the skin [6].
In vitro studies show CBDV can activate the vanilloid (TRPV1) receptors which are involved with pain transmission and temperature regulation [22]. It’s thought CBDV may offer painkilling benefits through this effect.
The benefits of CBDV include:
- May offer neuroprotective benefits
- Alleviates pain
- May reduce the severity of epileptic seizures
- Blocks acne formation
3. CBC (Cannabichromene)
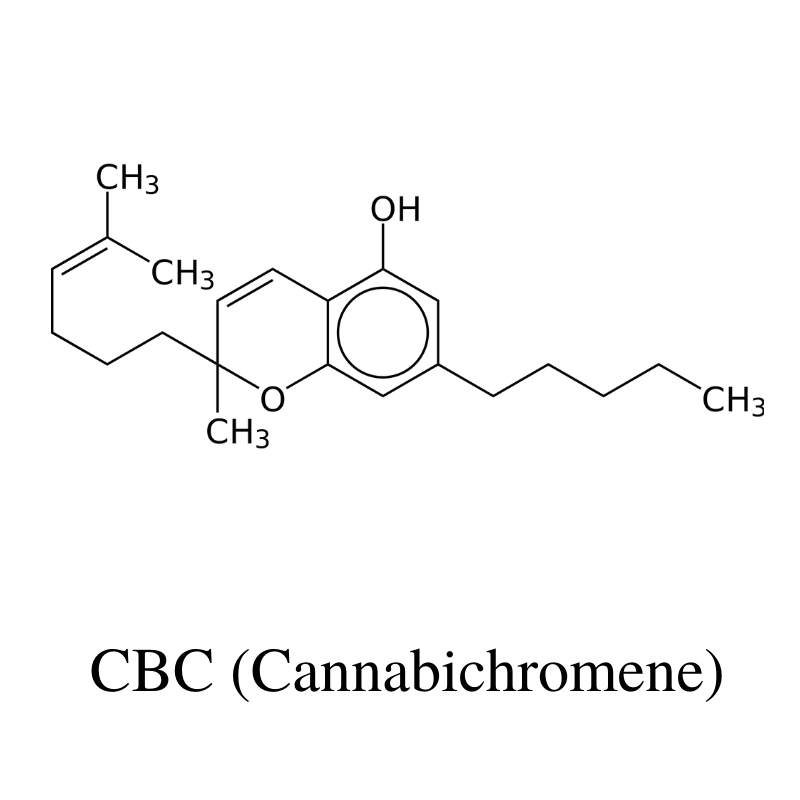
CBC (cannabichromene) is the third most abundant cannabinoid behind THC and CBD. It’s completely non-psychoactive like CBD but has a slightly different set of benefits.
This cannabinoid is much less studied than CBD, but there’s some evidence to suggest it is a promising candidate for inhibiting the growth of cancer cells [3], alleviating pain [4], and protecting neurons in the brain [5].
Other studies have shown CBC may reduce sebum to inhibit the formation of acne [6].
CBC has also been shown to have significant antidepressant effects in mice at a dose of 20 mg/kg [7].
The benefits of CBC include:
- Alleviates pain
- Protects brain & nerve cells
- May regulate mood & alleviate symptoms of depression
- Supports relaxation & sleep
- May slow the growth of cancer cells
- Prevents acne formation
4. CBCV (Cannabichromevarin)
CBCV has a similar structure and effect profile as CBC. Unfortunately, there’s very little information about the effects of this cannabinoid, and the majority of research on the effects of CBCV is theoretical in nature.
There’s been some research to suggest CBCV has anticonvulsant and anti-cancer benefits [23] — but more research is needed to confirm this effect.
The benefits of CBCV include:
- May slow the growth of cancer cells
- May support epileptic seizures
5. CBG (Cannabigerol)
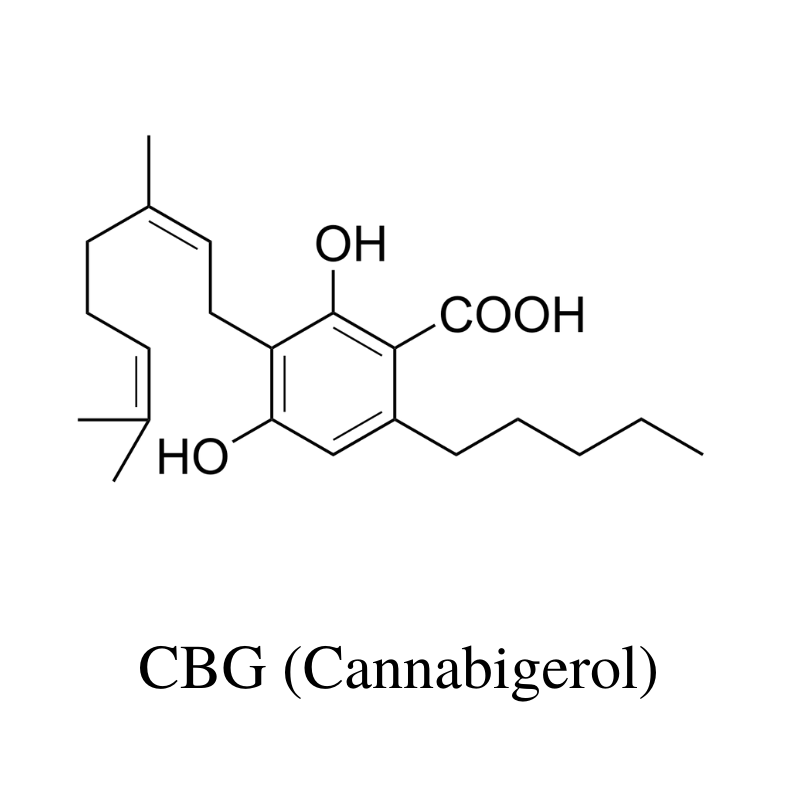
CBG (cannabigerol) is the precursor for CBD, THC, and CBC. It’s often referred to as the cannabinoid stem cell.
Most cannabis strains produce low concentrations of CBG (less than 1%). However, cannabis plants harvested before they’re completely ripe contain much more CBG than normal.
This cannabinoid has become a popular focus among health scientists for its unique health benefits. CBG oils, CBG concentrates, and CBG flowers are all becoming more common in the marketplace as well.
One of the most important benefits of CBG is its effects on a medical condition affecting the eye called glaucoma. This condition involves an increase in intraocular pressure causing pain. CBG has been shown to reduce intraocular pressure to ease pain [8].
Animal studies have shown CBG can protect the integrity of the skin suffering from various dry-skin syndromes [6].
Early animal studies have shown that CBG inhibits the growth of colon cancer in mice [9], stimulates appetite [10], offers neuroprotective effects against Huntington’s disease [11], and protects against inflammatory bowel disease [12].
The benefits of CBG include:
- Stimulates the appetite
- Relieves anxiety
- Protects nerve & brain cells
- May protect against cancer
- Inhibits the psychoactive effects of THC
- Relieves symptoms of glaucoma
- Inhibits bacterial growth
- Reduces inflammation
- May alleviate dry-skin syndrome
6. CBGM (Cannabigerol Monomethyl Ether)
Very little is known about CBGM — which shares a base structure of the more prominent cannabinoid, CBG.
Although the base structure is similar to CBG, this compound is classified as an ether — which could give it a very different set of effects compared to the more well-known CBG molecule.
CBGM was discovered in 1968, while a group of Japanese researchers was studying the chemical composition of a cannabis strain by the name of Minamiashigara No. 1 [24].
The benefits of CBGM include:
- May offer neuroprotective benefits
- May stimulate appetite
- May offer supportive effects against epileptic seizures
7. CBGV (Cannabigerovarin)
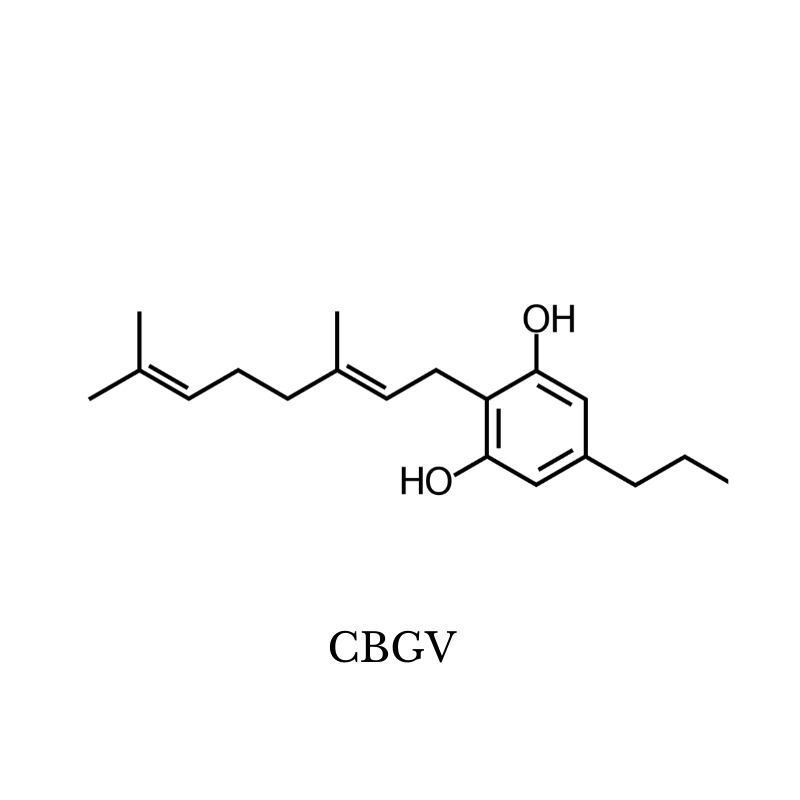
There isn’t much known about the therapeutic effects of CBGV (Cannabigerivarin), but research is beginning to pick up.
Here’s what we know about CBGV so far.
CBGV may offer analgesic effects through its ability to stimulate the vanilloid (TRPV1) receptors [25] — which are intimately involved in pain transmission.
Animal studies have shown CBGV increases sebum production in the skin and may offer supportive effects for conditions involving dry skin [6]. Other studies have shown CBGV may protect the gastrointestinal tract from inflammatory disorders such as Crohn’s disease or ulcerative colitis [26].
The benefits of CBGV include:
- May relieve dry skin conditions
- May protect the digestive tract from inflammatory conditions
8. CBL (Cannabicyclol)
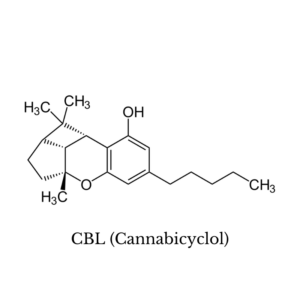
Very little is known about the effects of cannabicyclol. To date, the only studies that have been done on this compound have been focused on mapping out its chemical structure.
Interestingly, a sample of cannabis found in a tomb in central Asia contained this cannabinoid along with CBN as the predominant cannabinoids in the sample [27].
9. CBN (Cannabinol)
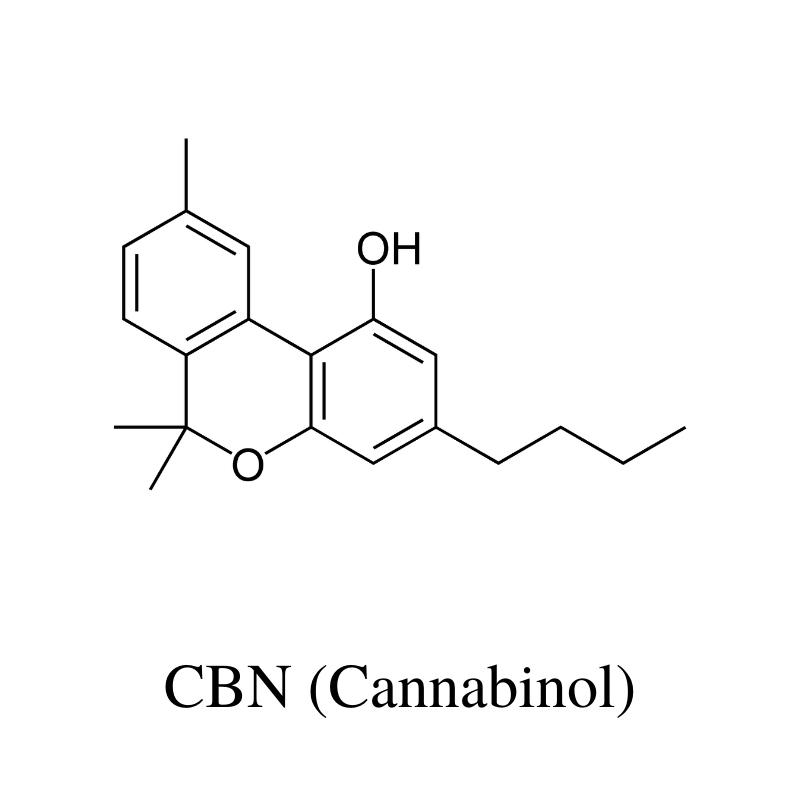
CBN (cannabinol) is a metabolite of THC ages and breaks down. Plants that are overripe or have been stored for long periods of time have the highest CBN content.
This cannabinoid is non-psychoactive but has a strong sedative action. Popular belief considers this cannabinoid as the primary compound that contributes to the sedative “couch-lock” effect in hemp or marijuana strains.
Dr. Ethan Russo, a prominent researcher in the cannabis space, suggests otherwise. He says older research conducted in the 70s got it wrong. CBN isn’t as sedating as previously thought — the sedative effects were more likely a result of other cannabis-derivatives in high CBN strains (such as terpenes).
Animal studies have shown CBN may offer neuroprotective effects against ALS [13], inhibit bacterial growth [14], stimulate appetite [15], alleviate inflammation associated with arthritis [16], and reduce intraocular pressure from glaucoma [17].
The benefits of CBN include:
- May slow the progression of ALS
- Protects brain & nerve cells
- Alleviates glaucoma pain
- Inhibits bacterial growth & infection
- Stimulates appetite
- Reduce inflammation
- Alleviate arthritis pain & inflammation
- May promote sleep
10. CBV (Cannabivarin)
CBV is an analog of CBN, which means it shares the same structure, but in a mirror image.
The effects of CBV aren’t well studied, but many experts suggest the effects are similar to CBN due to the resemblance in the shape of these molecules.
CBV is a byproduct of THCV as it’s broken down from exposure to heat or oxygen.
11. Delta 9 THC (Δ9-Tetrahydrocannabinol)
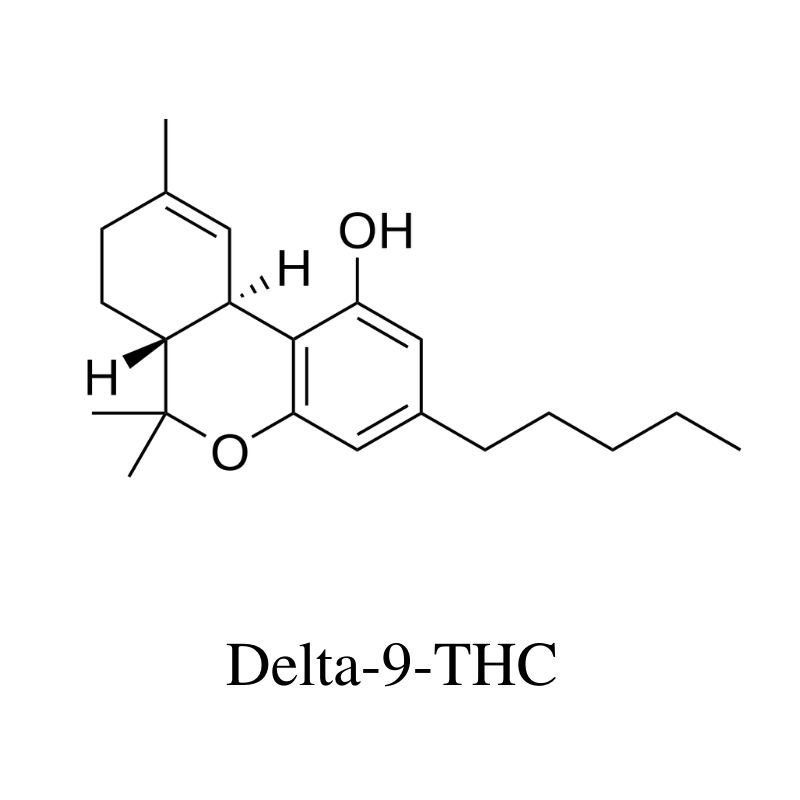
Delta 9 THC is the primary psychoactive cannabinoid in the marijuana plant. It works by stimulating the endocannabinoid receptors, some of which trigger a release of serotonin in the brain. It’s these effects on serotonin that make users high.
The prevalence of delta 9 THC is what determines the differences between hemp and marijuana plants.
Any cannabis plant that makes more than 0.3% THC is considered marijuana, while anything with 0.3% or less is hemp. The regulations are very different, depending on whether a plant is hemp or marijuana. Marijuana is restricted in most parts of the world.
In small doses (less than 15 mg), delta 9 THC is a mild sedative [28], while higher doses have a stimulating action [29].
The benefits of delta 9 THC include:
- Appetite stimulant
- Sedative (low doses)
- Stimulant (high doses)
- Produces a euphoric sensation
- Inhibits inflammation
- May relax the muscles
- Alleviates pain from glaucoma
- Reduces feelings of nausea
12. Delta 8 THC (Δ8-Tetrahydrocannabinol)
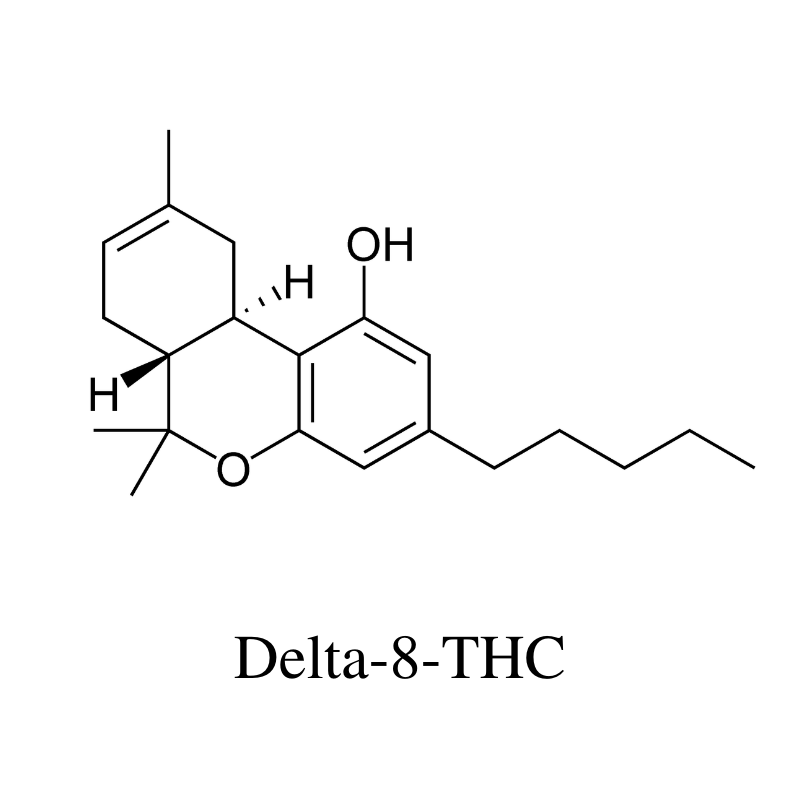
Delta 8 THC is an analog of delta 9 THC. In chemistry, an analog refers to a slightly different version of the same molecule. In the case of delta 8 THC and delta 9 THC, the difference is the location of a double bond — other than this, both compounds are identical.
Despite how similar these two chemicals are in terms of structure, they have some key differences in how they interact with the body.
Delta 8 THC has roughly half the psychoactivity as delta 9 THC and tends to produce a much more clear-headed buzz. It’s a popular alternative to delta 9 THC because it’s legal in many more parts of the world (and the United States), and gives a similar high without causing some of the negative side effects like anxiety or paranoia.
The cannabis plant doesn’t actually make delta 8 THC. There are no enzymes in the plant that manufacture this cannabinoid. Instead, the plant makes delta 9 THC, which can degrade over time to form delta 8 instead. Because of this, there are no plants that make high concentrations of delta 8 THC.
On the other hand, delta 8 THC can be made from CBD with the help of some chemical equipment.
The benefits of delta 8 THC include:
- Appetite stimulant
- Mild euphoric
- Clear-headed high sensation
- Inhibits inflammation
- Alleviates chronic pain
A similar cannabinoid, called delta 10 THC, offers the opposite effects. It produces more of the stimulating effects of THC and is active at around the same dose as delta 8.
13. THCV (Tetrahydrocannabivarin)
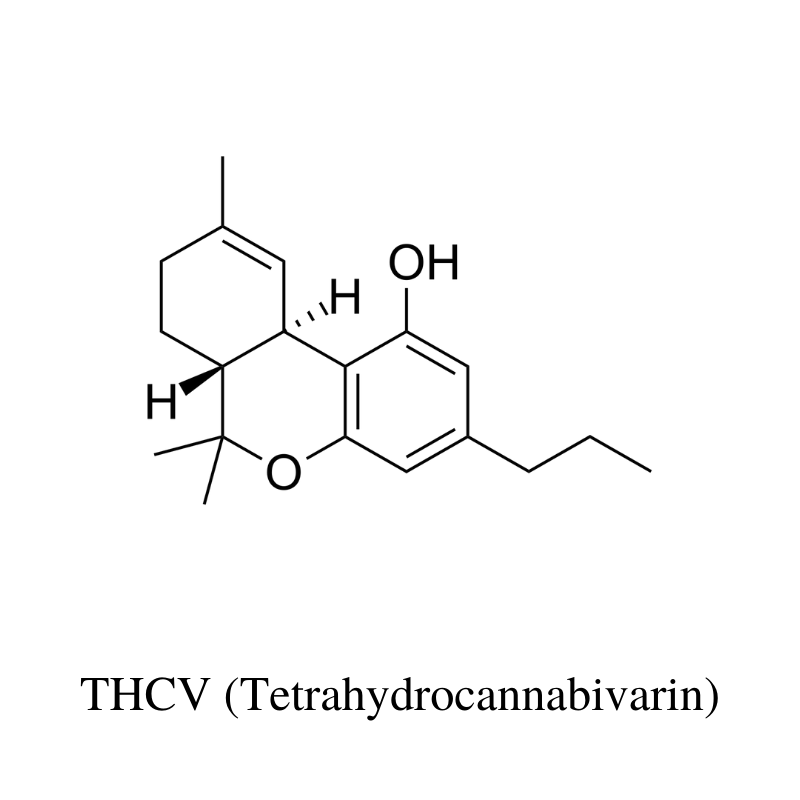
THCV (tetrahydrocannabivarin) is considered a minor cannabinoid because most strains of hemp and marijuana contain only trace amounts.
However, recent interest in this low-psychoactive cannabinoid has prompted breeders and cannabis extract companies to develop high-THCV strains and concentrates.
What’s the difference between THC and THCV?
Structurally, the only thing differentiating THC from THCV is two carbon atoms. Aside from this, THC and THCV are very different.
THCV is not only non-psychoactive but it’s also been shown to reduce THC’s psychoactive effects [18]. This cannabinoid inhibits some of the effects of THC (such as increased heart rate) and boosts others (anticonvulsant activity).
Animal studies have shown THCV may reduce insulin sensitivity to support diabetes [19], offer protective effects against neurological disorders such as Parkinson’s disease [20], and prevent acne formation [6].
Early in vitro research shows THCV stimulates bone and collagen formation [21], making it especially useful for bone and joint diseases such as osteoporosis or arthritis.
The benefits of THCV include:
- Reduces psychoactive effects of THC
- May reduce insulin-resistance
- May protect against Parkinson’s disease
- Prevents acne formation
- Reduces appetite
- Protects the brain & nerve cells
14. THCC (Tetrahydrocannabiorcol)
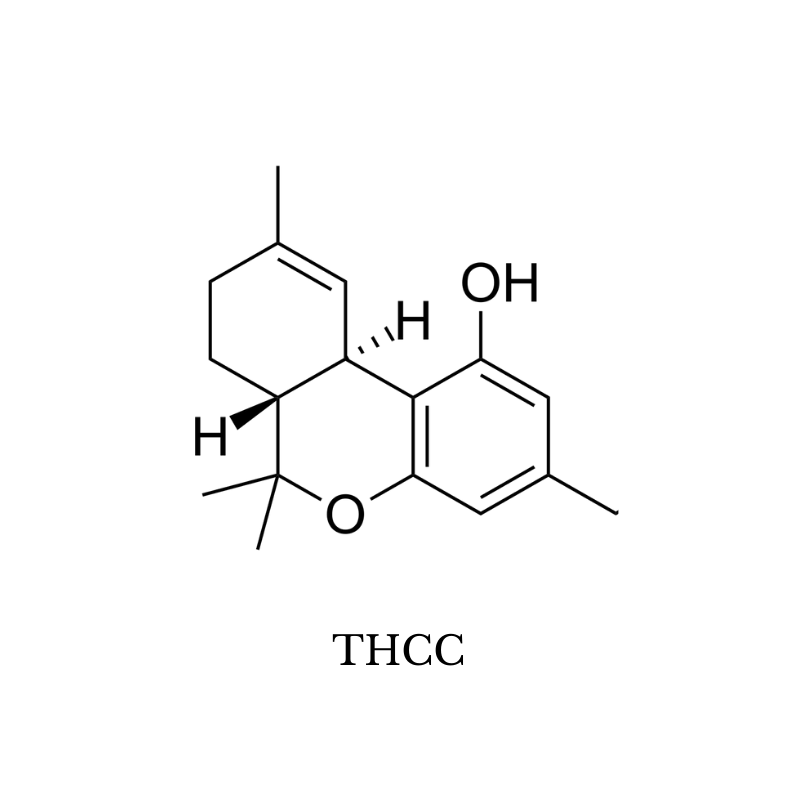
THCC (tetrahydrocannabiorcol) is one of the few forms of THC that has no psychoactive effects. Its effect profile is much closer to CBD than it is to THC.
This cannabinoid has just one carbon atom in its alkyl side chain. This length of this side chain heavily influences the potency of a given cannabinoid because it affects how easily the molecule is able to bind to the CB1 receptors (which is what causes the euphoric high associated with THC).
THCC has just one carbon and therefore has a low affinity for the CB1 receptors (non-psychoactive).
THCV has three carbons and has a mild affinity (about 25% the psychoactivity of THC).
Delta 7, 8, 9, and 10 THC all have five carbons. Delta 9 THC is the strongest of this group.
THCP has seven carbons and has roughly 33 times the potency of THC on the CB1 receptors.
Anything beyond 7 carbons isn’t found in nature.
While THCC doesn’t have a strong action on the endocannabinoid receptors, it does have a strong effect on the. TRP receptors — more specifically, the TRPA1, TRPV1, TRPV2, TRPV3, and TRPV4 ion channel receptors. The TRP ion channels (sometimes called receptors), act as chemical sensors.
TRP channels trigger an inflammatory and pain response when noxious chemicals, freezing cold, or high heat are detected. They’re intimately involved with pain transmission, certain kinds of inflammation, and are thought to be at least partly involved in the pathophysiology of diabetic neuropathy, autism spectrum disorder, migraine headaches, and muscular dystrophy.
The benefits of THCC include:
- May alleviate chronic neuropathy
- May help reduce increased pain sensitivity
- May support symptoms of autism spectrum disorder
15. HHC (Hydroxyhexahydrocannabinol)
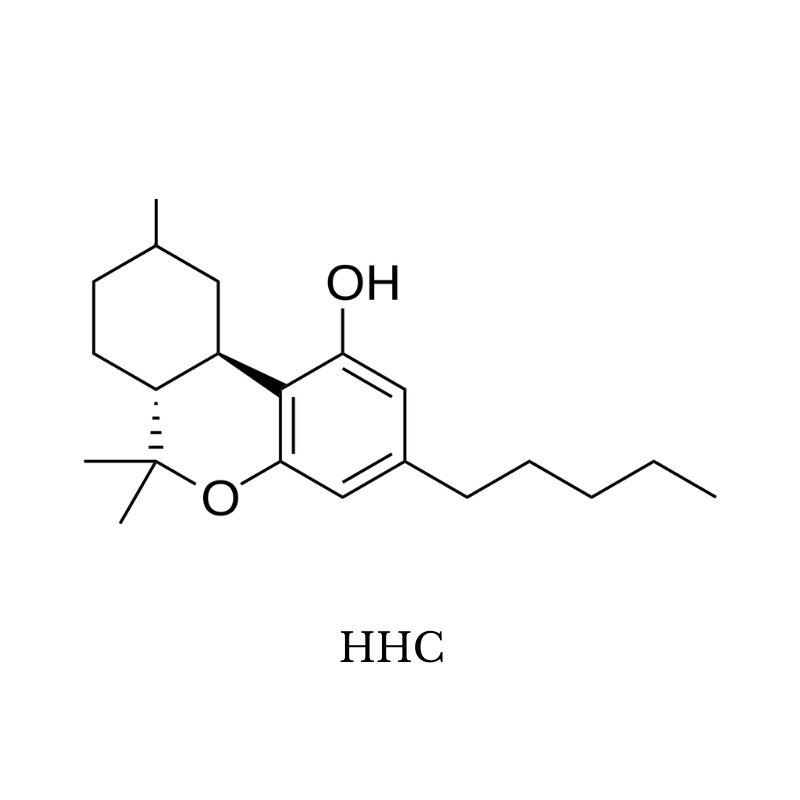
HHC stands for hydroxyhexahydrocannabinol — it’s a naturally occurring, but exceedingly rare version of THC.
This cannabinoid shares the same base structure as delta 8, 9, or 10 THC, but doesn’t contain any double bonds in its top ring structure.
As a reminder, delta 8, 9, and 10 THC are named according to the location of this double bond (delta 8 has a double bond on the eighth carbon, for example).
Instead of a double bond, all carbons in the structure are filled with hydrogen atoms which extend off the molecule in all directions. Because of this, it’s considered to be “hydrogenated” or “saturated.”
HHC is essentially the margarine of THC.
The effects of this cannabinoid are virtually identical as delta 9 THC — albeit just slightly weaker (around 80% the potency of delta 9 THC and slightly stronger than delta 8 THC).
The main benefit of this cannabinoid is its exceptionally long shelf-life. In its hydrogenated form, HHC is exceptionally stable — resistant to UV radiation, oxygen, and heat. It’s unclear exactly how long HHC will remain viable — but early research suggests it’s significantly longer than conventional THC — which is the least stable of them all.
For you preppers out there, this is the cannabinoid you should be adding to your doomsday stockpile.
The Benefits of HHC Include:
- Appetite stimulant
- Sedative (low doses)
- Stimulant (high doses)
- Produces a euphoric sensation
- Inhibits inflammation
- May relax the muscles
- Alleviates pain from glaucoma
- Reduces feelings of nausea
16. THCP
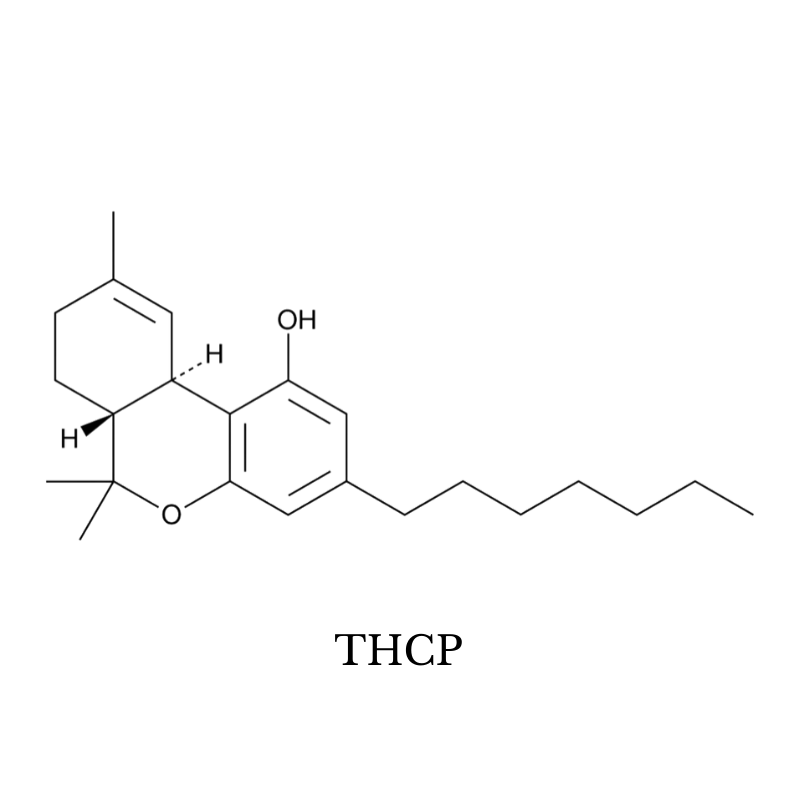
THCP (tetrahydrocannabiphorol) is the strongest naturally occurring cannabinoid known. It’s been shown to have nearly 33 times the potency of delta 9 THC on the CB1 receptors.
Whether this translates to 33 times the potency is up for debate. Most people who have used it place the potency somewhere between 5 and 10 times that of delta 9 THC. Either way, this stuff is exceptionally strong — nearly psychedelic in its effects.
This cannabinoid can be found in marijuana plants. In fact, it was first discovered while a team of Italian researchers was investigating a mysterious spike on a mass spectrometry chart for a particularly strong strain called FM2.
Some experts suggest THCP could be the reason why some strains of marijuana are so much stronger than others despite having the same or less THC content. Even sub-milligram doses of this cannabinoid are psychoactive and would add to the overall effect profile.
II. Endocannabinoids
Endo- refers to compounds “inside” the body. These compounds are produced by the body’s own cells to interact with the endocannabinoid system.
There are two main endocannabinoids (anandamide and 2-AG) that are responsible for the majority of activity in the ECS. Others, such as OAE, NADA, and LPI, make up a smaller percentage of activity within specific tissues.
Endocannabinoids are classified as eicosanoids — which is a class of compounds made from arachidonic acid. The body makes many different eicosanoids, which it uses as chemical messengers to drive inflammation, allergies, immune activity, pain transmission, cell growth, reproductive cycles, and more.
Anandamide
Anandamide, also known as arachidonoylethanolamine, is considered the primary regulator of the endocannabinoid system (ECS). It’s found in every tissue type in the body but has the highest concentration in organs outside the brain.
The name, anandamide, comes from the Sanskrit word for bliss — amide. It’s often referred to as the “bliss” molecule and is thought to play a role in producing feelings of contention and satisfaction.
2-AG
2-AG stands for 2-arachidonoylglycerol.
It’s the second primary endocannabinoid in the body and makes up the majority of the endocannabinoid activity in the brain. This endocannabinoid is a full-agonist for both the CB1 and CB2 receptors.
Virodhamine (OAE)
OAE stands for O-arachidonoyl-ethanolamine (OAE).
This lesser cannabinoid is much less studied than anandamide or 2-AG. So far, it’s been found to act as a full-agonist for the CB2 receptors and a partial agonist for CB1 receptors.
N-Arachidonoyl Dopamine (NADA)
NADA is a lesser endocannabinoid with similar activity to anandamide. It binds to the CB1 receptors as well as vanilloid (TRPV1 receptors).
Lysophosphatidylinositol (LPI)
This compound hasn’t been officially confirmed as an endocannabinoid. However, early research suggests it binds to the GPR55 endocannabinoid receptors.
More research is needed to understand the effects of this compound and its role in the endocannabinoid system.
III. Synthetic Cannabinoids
Synthetic cannabinoids share a similar structure to naturally occurring cannabinoids but are manufactured in a lab. The most popular synthetic cannabinoid available today is THC-O — which is roughly 3 times as potent as conventional delta 9 THC.
Most synthetic cannabinoids are structurally similar to THC. There was a boom in the synthetic cannabinoid space in the early 2010s as manufacturers were on the hunt for products they could sell as a “legal high.” At the time, marijuana products were illegal, but synthetic derivatives weren’t. Products containing these synthetic THC compounds were sold in headshops and online as a “legal” high.
Synthetic cannabinoids were sold under the common names “spice” or “K2”. Spice was made by spraying dried leaves with a blend of synthetic cannabinoids. These products were then smoked like marijuana.
These compounds were notorious for their side effects, which range from severe headaches, lung damage, or seizures.
Today, marijuana is legal in many parts of the world, and all psychoactive substances are illegal until proven safe as per the New Psychoactive Substances Act of 2013 (USA). Spice is only available on the black market today but remains unpopular due to the negative side effects, high level of risk, and ready availability of marijuana.
Synthetic cannabinoids don’t always follow the same structure as natural cannabinoids. True cannabinoids are classified as eicosanoids — which use arachidonic acid as the base. You can find synthetic cannabinoids in the form of aminoalkylindoles, 1,5-diaryl pyrazoles, quinolines, and aryl sulfonamides — which are very different than naturally occurring cannabinoids but interact with the same receptors.
List of Synthetic Cannabinoids:
- THC-O-Acetate
- JWH-018
- JWH-073
- JWH-200
- AM-2201
- UR-144
- XLR-11
- AKB4
- Cannabicyclohexanol
- AB-CHMINACA
- AB-PINACA
- AB-FUBINACA
IV. Pharmaceutical Cannabinoids
Over the past few years, there’s been a big push in the pharmaceutical space to develop new cannabinoid-based drugs.
Several large pharmaceutical companies such as GW Pharmaceuticals, Solvay Pharmaceuticals, Pharmos, and Valeant Pharmaceuticals are investing heavily in the “cannabinoids as medicine” space.
Sativex®
Sativex is one of the most well-known and researched pharmaceutical cannabis preparations. It’s made by the British Pharmaceutical giant, GW Pharmaceuticals. It’s used in the treatment of pain from multiple sclerosis and cancer.
This medication comes in the form of an oral spray with a 1:1 preparation of THC and CBD.
Sativex uses natural sources of CBD and THC rather than synthetic versions like many of the other pharmaceutical options on this list.
Epidiolex®
Epidiolex is the only FDA-approved CBD product for the treatment of epileptic disorders (Dravet’s syndrome and Lennox-Gastaut Syndrome). It’s also used to treat a condition called Tuberous Sclerosis Complex.
This product is made by the pharmaceutical giant Greenwich Biosciences — a subsidiary of GW Pharmaceuticals that specializes in the development of cannabinoid-based medicines in the United States.
Dexanabinol®
Dexanabinol is a synthetic cannabinoid, but it doesn’t interact with the endocannabinoid system. Instead, this compound binds to the NMDA glutamate receptors, where it’s used to reduce convulsions and offer broad neuroprotective benefits.
This product is made by the American pharmaceutical company, Pharmos.
Marinol®
Marinol is a synthetic version of delta-9-THC made by Unimed Pharmaceuticals (a subsidiary of Solvay Pharmaceuticals).
This FDA-approved medication is used to manage vomiting in cancer patients, as a painkiller for multiple sclerosis, and for stimulating appetite in AIDS patients.
Cesamet®
Cesamet is a synthetic derivative of THC. It’s similar in shape and effects, but the structure is slightly different from THC.
This drug is made by Valeant Pharmaceuticals International for treating side effects of cancer therapy — such as vomiting and neuropathic pain.
This medication was one of the first on the market, earning approval from the FDA back in 1985.
Cannabinor
This pharmaceutical was developed by Pharmos, who is also the manufacturer of Dexanabinol.
Cannabinor is a synthetic cannabinoid that binds exclusively to the CB2 endocannabinoid receptors. It’s used to reduce inflammation, improve bladder control, and alleviate chronic pain.
CT-3
CT-3 is a synthetic cannabinoid modeled after a metabolite of THC — THC-11-oic acid. It’s used to treat neuropathic pain and muscle spasticity in multiple sclerosis patients.
This product is made by Indevus Pharmaceuticals.
Taranabant
This synthetic molecule binds to the CB1 endocannabinoid receptors but causes them to do the opposite effect (inverse agonist). It’s used to reduce appetite as a potential weight-loss treatment.
This drug never made it through phase III clinical trials. The creator, Merck, stopped development after finding too many patients were experiencing side effects, including anxiety and depression.
V. Cannabinoids In Other Plant Species
While cannabis is the king of cannabinoids, there are several other species that produce compounds that can interact with the endocannabinoid system as well.
Here are a few other species of plants that contain cannabinoids:
- Echinacea purpurea (Purple coneflower) — produce compounds called N-alkylamides, which have been shown to interact with the CB2 endocannabinoid receptors
- Acmella oleracea (Electric daisy) — produces a group of compounds called N-isobutylamides, which stimulate the CB2 receptors
- Piper methysticum (Kava kava) — one of the active ingredients, yangonin, interacts with the CB1 endocannabinoid receptors
- Camellia sinensis (green tea plant) — catechins in the tea plant have an affinity for endocannabinoid receptors
- Tuber melanosporum (Black truffle) — contains the endocannabinoid anandamide
- Radula marginata (Liverwort) — contains a cannabinoid called perrottetinene, which is very similar in structure to THC
Key Takeaways: What Are Cannabinoids?
Cannabinoids are a large and diverse group of chemicals that interact with the endocannabinoid system — which is found in all of our internal organs, including the skin, blood vessels, and immune system.
Cannabinoids that are produced by our own body are called endocannabinoids, and the cannabinoids made by plants like hemp are called phytocannabinoids.
These compounds offer an impressive range of benefits that range from regulating our sleep, hormones, pain transmission, inflammation, appetite, and much more.
The most common cannabinoids are THC (in marijuana), CBD (in hemp), and CBG. They’re popular health supplements for supporting everything from chronic pain and inflammation to mood disorders like anxiety or depression.
References
- Aizpurua-Olaizola, O., Soydaner, U., Öztürk, E., Schibano, D., Simsir, Y., Navarro, P., … & Usobiaga, A. (2016). Evolution of the cannabinoid and terpene content during the growth of Cannabis sativa plants from different chemotypes. Journal of natural products, 79(2), 324-331.
- Mechoulam, R., Peters, M., Murillo‐Rodriguez, E., & Hanuš, L. O. (2007). Cannabidiol–recent advances. Chemistry & biodiversity, 4(8), 1678-1692.
- Ligresti, A., Moriello, A. S., Starowicz, K., Matias, I., Pisanti, S., De Petrocellis, L., … & Di Marzo, V. (2006). Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. Journal of Pharmacology and Experimental Therapeutics, 318(3), 1375-1387.
- Maione, S., Piscitelli, F., Gatta, L., Vita, D., De Petrocellis, L., Palazzo, E., … & Di Marzo, V. (2011). Non‐psychoactive cannabinoids modulate the descending pathway of antinociception in anaesthetized rats through several mechanisms of action. British journal of pharmacology, 162(3), 584-596.
- Shinjyo, N., & Di Marzo, V. (2013). The effect of cannabichromene on adult neural stem/progenitor cells. Neurochemistry international, 63(5), 432-437.
- Oláh, A., Markovics, A., Szabó‐Papp, J., Szabó, P. T., Stott, C., Zouboulis, C. C., & Bíró, T. (2016). Differential effectiveness of selected non‐psychotropic phytocannabinoids on human sebocyte functions implicates their introduction in dry/seborrhoeic skin and acne treatment. Experimental dermatology, 25(9), 701-707.
- El-Alfy, A. T., Ivey, K., Robinson, K., Ahmed, S., Radwan, M., Slade, D., … & Ross, S. (2010). Antidepressant-like effect of Δ9-tetrahydrocannabinol and other cannabinoids isolated from Cannabis sativa L. Pharmacology Biochemistry and Behavior, 95(4), 434-442.
- Nadolska, K., & Goś, R. (2008). Possibilities of applying cannabinoids’ in the treatment of glaucoma. Klinika oczna, 110(7-9), 314-317.
- Borrelli, F., Pagano, E., Romano, B., Panzera, S., Maiello, F., Coppola, D., … & Izzo, A. A. (2014). Colon carcinogenesis is inhibited by the TRPM8 antagonist cannabigerol, a Cannabis-derived non-psychotropic cannabinoid. Carcinogenesis, 35(12), 2787-2797.
- Brierley, D. I., Samuels, J., Duncan, M., Whalley, B. J., & Williams, C. M. (2017). A cannabigerol-rich Cannabis sativa extract, devoid of [INCREMENT] 9-tetrahydrocannabinol, elicits hyperphagia in rats. Behavioural pharmacology, 28(4), 280-284.
- Valdeolivas, S., Navarrete, C., Cantarero, I., Bellido, M. L., Muñoz, E., & Sagredo, O. (2015). Neuroprotective properties of cannabigerol in Huntington’s disease: studies in R6/2 mice and 3-nitropropionate-lesioned mice. Neurotherapeutics, 12(1), 185-199.
- Borrelli, F., Fasolino, I., Romano, B., Capasso, R., Maiello, F., Coppola, D., … & Izzo, A. A. (2013). Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochemical pharmacology, 85(9), 1306-1316.
- Weydt, P., Hong, S., Witting, A., Möller, T., Stella, N., & Kliot, M. (2005). Cannabinol delays symptom onset in SOD1 (G93A) transgenic mice without affecting survival. Amyotrophic Lateral Sclerosis, 6(3), 182-184.
- Appendino, G., Gibbons, S., Giana, A., Pagani, A., Grassi, G., Stavri, M., … & Rahman, M. M. (2008). Antibacterial cannabinoids from Cannabis sativa: a structure− activity study. Journal of natural products, 71(8), 1427-1430.
- Farrimond, J. A., Whalley, B. J., & Williams, C. M. (2012). Cannabinol and cannabidiol exert opposing effects on rat feeding patterns. Psychopharmacology, 223(1), 117-129.
- Zurier, R. B., & Burstein, S. H. (2016). Cannabinoids, inflammation, and fibrosis. The FASEB Journal, 30(11), 3682-3689.
- Kogan, N. M., & Mechoulam, R. (2007). Cannabinoids in health and disease. Dialogues in clinical neuroscience, 9(4), 413.
- Englund, A., Atakan, Z., Kralj, A., Tunstall, N., Murray, R., & Morrison, P. (2016). The effect of five day dosing with THCV on THC-induced cognitive, psychological and physiological effects in healthy male human volunteers: a placebo-controlled, double-blind, crossover pilot trial. Journal of Psychopharmacology, 30(2), 140-151.
- Wargent, E. T., Zaibi, M. S., Silvestri, C., Hislop, D. C., Stocker, C. J., Stott, C. G., … & Cawthorne, M. A. (2013). The cannabinoid Δ 9-tetrahydrocannabivarin (THCV) ameliorates insulin sensitivity in two mouse models of obesity. Nutrition & diabetes, 3(5), e68-e68.
- Garcia, C., Palomo‐Garo, C., García‐Arencibia, M., Ramos, J. A., Pertwee, R. G., & Fernández‐Ruiz, J. (2011). Symptom‐relieving and neuroprotective effects of the phytocannabinoid Δ9‐THCV in animal models of Parkinson’s disease. British journal of pharmacology, 163(7), 1495-1506.
- Scutt, A., & Williamson, E. M. (2007). Cannabinoids stimulate fibroblast colony formation by bone marrow cells indirectly via CB 2 receptors. Calcified Tissue International, 80(1), 50-59.
- Iannotti, F. A., Hill, C. L., Leo, A., Alhusaini, A., Soubrane, C., Mazzarella, E., … & Stephens, G. J. (2014). Non Psychotropic plant cannabinoids, cannabidivarin (CBDV) and cannabidiol (CBD), activate and desensitize transient receptor potential vanilloid 1 (TRPV1) channels in vitro: potential for the treatment of neuronal hyperexcitability. ACS chemical neuroscience, 5(11), 1131-1141.
- Stone, N. L., Murphy, A. J., England, T. J., & O’Sullivan, S. E. (2020). A systematic review of minor phytocannabinoids with promising neuroprotective potential. British Journal of Pharmacology, 177(19), 4330-4352.
- Yamauchi, T., Shoyama, Y., Matsuo, Y., & Nishioka, I. (1968). Cannabigerol monomethyl ether, a new component of hemp. Chemical and Pharmaceutical Bulletin, 16(6), 1164-1165.
- De Petrocellis, L., Ligresti, A., Moriello, A. S., Allarà, M., Bisogno, T., Petrosino, S., … & Di Marzo, V. (2011). Effects of cannabinoids and cannabinoid‐enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. British journal of pharmacology, 163(7), 1479-1494.
- De Petrocellis, L., Orlando, P., Moriello, A. S., Aviello, G., Stott, C., Izzo, A. A., & Di Marzo, V. (2012). Cannabinoid actions at TRPV channels: effects on TRPV3 and TRPV4 and their potential relevance to gastrointestinal inflammation. Acta physiologica, 204(2), 255-266.
- Russo, E. B., Jiang, H. E., Li, X., Sutton, A., Carboni, A., Del Bianco, F., … & Zhang, Y. B. (2008). Phytochemical and genetic analyses of ancient cannabis from Central Asia. Journal of experimental botany, 59(15), 4171-4182.
- Nicholson, A. N., Turner, C., Stone, B. M., & Robson, P. J. (2004). Effect of Δ-9-tetrahydrocannabinol and cannabidiol on nocturnal sleep and early-morning behavior in young adults. Journal of clinical psychopharmacology, 24(3), 305-313.
- Evans, M. A., Harbison, R. D., Brown, D. J., & Forney, R. B. (1976). Stimulant actions of Δ 9-Tetrahydrocannabinol in mice. Psychopharmacology, 50(3), 245-250.
Signup to our newsletter
Be the first to know about our newest arrivals and special offers!